Orellanina
Orellanina
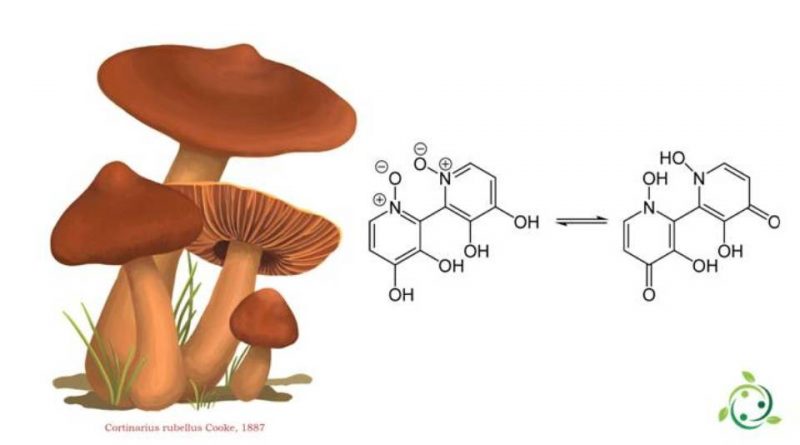
L’orellanina il cui termine nella nomenclatura ufficiale IUPAC è: 3,3 ′, 4,4′-Tetraidrossi-2,2′-bipiridina-N, N′-biossido, conosciuta anche con il nome di: 2,2-bipiridina-3,3-4,4-tetrol-1,1-diossido è un alcaloide.
L’orellanina ha formula bruta o molecolare: C10H8O6N2 e dal punto di vista fisico si presenta come un cristallo incolore.
L’orellanina è una micotossina citotossica contenuta in alcuni funghi del genere Cortinarius (Cortinarius orellanus Fr., 1838 e Cortinarius rubellus Cooke, 1887).
L’orellanina venne isolata per la prima volta nel 1955 dal medico polacco Stanisław Grzymała ma la struttura chimica è stata scoperta solo nell’ultima metà degli anni settanta dai chimici polacchi Antkowiak e Gessner.
L’orellannina ha una doppia struttura ad anello in cui entrambi gli anelli sono piridine (anelli eterociclici con un atomo di azoto). Parallelamente Antkowiak e Gessner scoprirono anche una sostanza simile, l’orellina che pur essendo affine dal punto di vista strutturale non è tossica.
L’orellanina è invece tossica ed è responsabile della sindrome orellanica.
L’orellanina, che viene estratta dai carpofori di Cortinarius orellanus ed altri Cortinarius è una sostanza cristallina simile allo zucchero ed è una bipiridina idrolizzata che non ha odori e sapori; è inattivata ad altissime temperature, e resa inoffensiva dalla luce. Per esposizione a luce U.V. o a temperature maggiori di 270° C, viene trasformata in orellina ed in orellinina.
L’orellanina si degrada in orellina, un composto che non presenta tossicità sugli animali.
L’orellanina è quindi abbastanza termostabile tanto che la tossicità dei carpofori non è alterata né con la cottura né con l’essiccamento, almeno fino a temperature di 150 °C. Nel giro di pochi giorni, l’orellanina, va a depositarsi nel rene in modo definitivo, costringendo il paziente ad effettuare per tutta la vita la dialisi, ed in molti casi il trapianto renale.
La dose minima letale (LD50), si avvicina a 50 g. di fungo fresco, sarebbero pertanto sufficienti 40-50 g. di fungo fresco per causare un avvelenamento mortale, in un soggetto adulto.
Vi è da dire però che la concentrazione di tossine riferite ad una determinata specie fungina, subisce cambiamenti a seconda delle varie aree geografiche ed esiste anche una variabilità individuale tra i vari soggetti, poiché non tutti gli intossicati vanno incontro ad insufficienza renale. Le cortinarine, strutturalmente simili alle amanitine, avrebbero un coinvolgimento nella sindrome orellanica; allo stato attuale delle ricerche non ci sono però conferme definitive.
L’attività citotossica di queste cortinarine, inibirebbe l’RNA-polimerasi e quindi la sintesi proteica; esse avrebbero la capacità di bloccare le funzioni renali con effetto cumulativo, e avrebbero un’azione ritardata.
La sintomatologia di assunzione di questo alcaloide è la seguente:
– I fase: dopo 4-9 ore compaiono secchezza della mucosa orale, sete insaziabile, urinamento continuo; quindi si passa a una seconda fase con mancanza di urina, vomito, diarrea, dolori addominali; infine la sindrome gastrointestinale regredisce rapidamente finché il soggetto va incontro ad un periodo di benessere (remissione);
– II fase: alla I fase segue la fase di insufficienza renale acuta (IRA), l’intossicazione non ben trattata provoca la morte per uremia;
Nel caso che il soggetto intossicato si salvi dovrà sottoporsi alla dialisi per tutta la vita o fino ad un eventuale trapianto di reni.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.