Ellagitannini
Ellagitannini
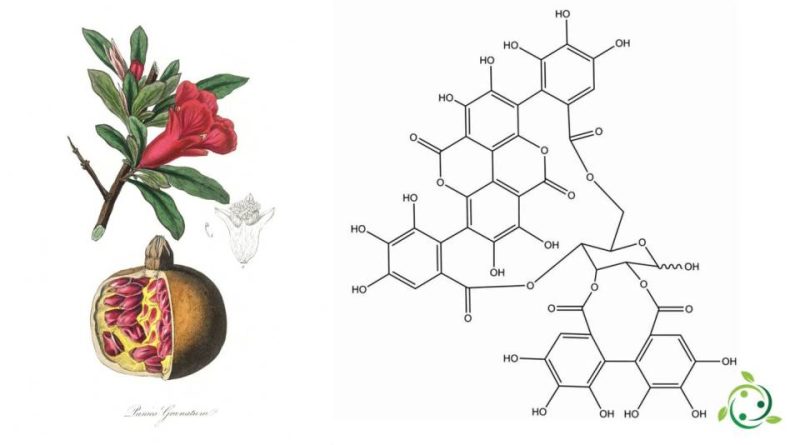
Gli ellagitannini sono una classe di tannini idrolizzabili, dei polifenoli formati principalmente dal legame ossidativo dei gruppi galloile nel glucosio 1,2,3,4,6-pentagalloile.
Dal punto di vista chimico gli ellagitannini differiscono dai gallotannini, in quanto i loro gruppi galloilici sono collegati tramite legami C-C, mentre i gruppi galloilici nei gallotannini sono collegati da legami depside (composto polifenolico costituito da due o più unità aromatiche).
Gli ellagitannini contengono un numero variabile di unità esaidrossidifenoiliche, nonché unità galloiliche e/o unità sanguisorboiliche legate alla frazione zuccherina.
Per determinare la quantità di ogni singola unità si esegue l’idrolisi degli estratti con acido trifluoroacetico in sistema metanolo/acqua. L’acido esaidrossidifenico, creato dopo l’idrolisi, viene spontaneamente lattonato ad acido ellagico e l’acido sanguisorbico ad acido sanguisorbico dilattone, mentre l’acido gallico rimane intatto.
Gli ellagitannini generalmente formano macrocicli, mentre i gallotannini no.
Gli ellagitannini sono presenti nel mondo vegetale come nella melagrana, frutto del melograno (Punica granatum L., 1753) che contiene numerose sostanze benefiche per il nostro organismo, tra cui ellagitannini, vitamine e minerali.
Gli ellagitannini sono molecole particolarmente interessanti poiché dotate di note proprietà antiossidanti e neuroprotettive e di una probabile azione anti tumorale.
Gli ellagitannini, oltre che nella melagrana sono presenti nelle noci e nei frutti di bosco: una volta idrolizzati a livello intestinale, gli ellagitannini liberano acido ellagico da cui deriva l’urolitina A.
Negli studi condotti in passato su animali anziani, l’urolitina A è risultata in grado di stimolare la degradazione selettiva dei mitocondri difettosi, e di migliorare la salute muscolare.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.