Disolfuro di diallile
Disolfuro di diallile
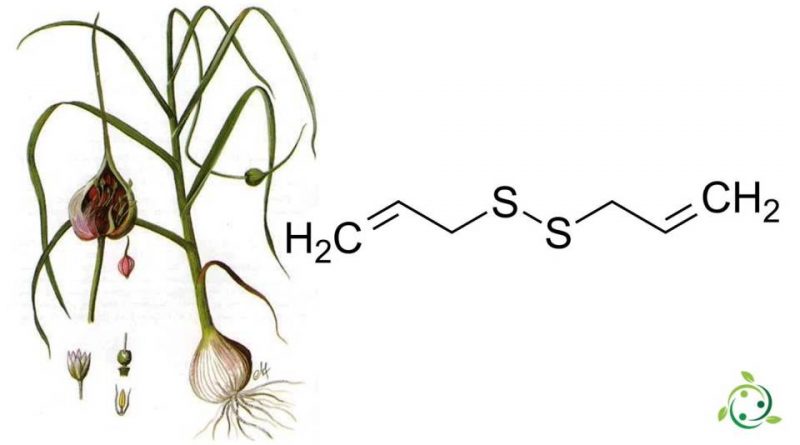
Il disolfuro di diallile, il cui termine nella nomenclatura ufficiale IUPAC è: 3-[(Prop-2-en-1-il)disulfanil]prop-1-ene ha formula bruta o molecolare: C6H10S2.
Il disolfuro di diallile è un composto chimico che si forma in maniera abbondante nell’estratto degli oli essenziali delle bulbose del genere Allium (come aglio, cipolla, porro, erba cipollina e scalogno).
Questa sostanza, dal punto di vista fisico, è un liquido irritante e infiammabile dal caratteristico odore d’aglio, liposolubile e quasi insolubile in acqua. La sua stabilità rispetto ai composti più ossigenati da cui naturalmente deriva lo fanno preferire nell’utilizzo nell’industria alimentare
Il disolfuro di diallile, unitamente ad alliina, allicina, ajoene, trisolfuro di diallile, tetrasolfuro di diallile, rientra nella composizione delle sostanze che danno, insieme agli oli essenziali terpenici, il caratteristico aroma alle bulbose.
In ambito biochimico le sostanze di partenza che conducono alla formazione dei solfuri allilici hanno origine da composti come alliina ed isoalliina che, sottoposti ad azione enzimatica da parte dell’allinasi, contenuta nel sistema di vacuoli cellulari della stessa pianta, li convertono nei sulfonati, passando per intermedi sulfenici. Queste reazioni avvengono sia nel distillato, sia nel macerato oleoso. A seconda delle condizioni di estrazione, può poi avvenire la riduzione a solfuro.
Il disolfuro di diallile oggetto di studi per quanto concerne le proprietà anti-tumorali dell’aglio ed in particolare in relazione all’azione sui carcinomi in genere.
In tal senso sono in corso studi sia in vivo ed in vitro per appurarne l’effettivo ruolo. Nello specifico si ritiene che l’attività del disolfuro di diallile si esplichi nell’arresto del ciclo cellulare in fase G2/M e nell’induzione di apoptosi nelle cellule tumorali.
Tuttavia l’aglio, unitamente ad altre bulbose, esplicherebbe la sua azione anche grazie ad un vasto spettro di composti potenzialmente protettivi, prevalentemente ad azione antiossidante.
Il disolfuro di diallile non era conosciuto prima del 1844 quando Theodor Wertheim separò mediante distillazione di vapore una sostanza dall’odore pungente dall’aglio e la chiamò “zolfo allilico”. Tuttavia, solo nel 1892 Friedrich Wilhelm Semmler riuscì a identificare il disolfuro di diallile come uno dei componenti dell’olio distillato di aglio. Il precursore naturale del disolfuro di diallile, l’allicina, fu scoperto nel 1944 da Chester J. Cavallito e John Hays Bailey. Nel 1947, A. Stoll ed E. Seebeck scoprirono che l’allicina a sua volta può essere prodotta dal derivato della cisteina alliina usando l’enzima alliinasi.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.