Aucubina
Aucubina
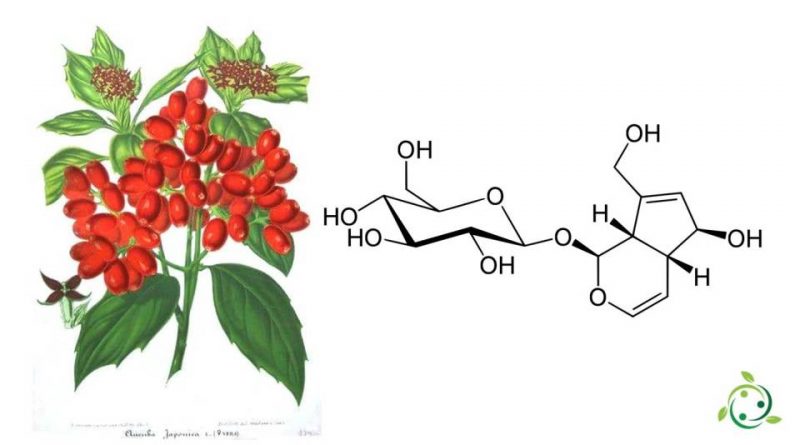
L’aucubina, il cui termine nella nomenclatura ufficiale IUPAC è: (1S,4aR,5S,7aS)-5-idrossi-7-(idrossimetil)-1,4a,5,7a-tetraidrociclopenta[c]piran-1-il β-D-glucopiranoside è un iridoide glicoside con formula bruta o molecolare: C15H22O9.
L’aucubina è un iridoide glicoside che è presente in natura in alcune piante come nelle foglie di Aucuba japonica (Cornaceae), Eucommia ulmoides (Eucommiaceae), e Plantago asiatica, Plantago major, Plantago lanceolata (Plantaginaceae), Galium aparine (Rubiaceae) ed altre ancora.
L’aucubina, come altri glicosidi iridoidi, svolge nelle piante il ruolo di composto di difesa e riduce i tassi di crescita di molti erbivori generalisti.
Si è scoperto che l’aucubina protegge dai danni al fegato indotti dal tetracloruro di carbonio o dall’alfa-amanitina nei topi e nei ratti quando 80 mg/kg sono stati somministrati per via intraperitoneale.
L’aucubina è, inoltre, un aperiente (farmaco a blanda azione purgativa) e stimola la secrezione di acido urico dai reni.
La biosintesi dell’aucubina vede il geranil pirofosfato (GPP) come precursore degli iridoidi. Il geranil fosfato viene generato attraverso la via del mevalonato o la via del metileritritolo fosfato.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.

Good afternoon
Only a question:
Is tha aucubin soluble in water or oils?
Thank you
Good morning, Aucubin, like all glycosides (aromatic organic compounds), is on average soluble in water (solubility in water = 50 g / L) and alcohol, slightly soluble in ether.