Acido Antranilico
Acido Antranilico
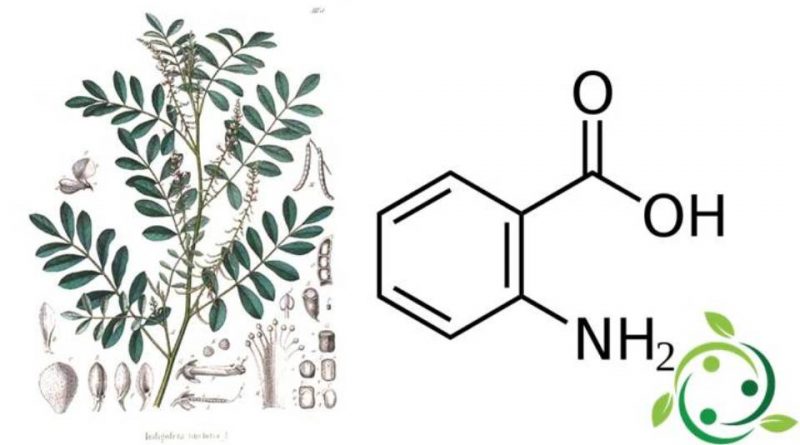
L’acido antranilico, il cui termine nella nomenclatura ufficiale IUPAC è: acido 2-amminobenzoico, conosciuto anche con i nomi alternativi di acido orto-amminobenzoico o acido o-amminobenzoico è un acido aromatico con formula bruta o molecolare: C7H7NO2.
L’acido antranilico, anche se di solito non viene indicato come tale, è un amminoacido. L’acido antranilico solido è costituito sia dall’acido ammino-carbossilico che dalle forme carbossilato di ammonio zwitterionico.
La molecola dell’ acido antranilico è costituita da un anello benzenico, orto-sostituito con un acido carbossilico e un’ammina.
Per via dei vari gruppi funzionali sia acidi che basici, il composto è anfotero.
L’acido antranilico, a temperatura ambiente si presenta come un solido giallo, inodore e poco solubile in acqua. Inoltre l’acido antranilico ha un sapore dolciastro.
L’anione ottenuto dalla deprotonazione dell’acido antranilico, è chiamato antranilato.
A livello industriale, l’acido antranilico è prodotto dall’anidride ftalica, a partire dall’amminazione:
C6H4 (CO) 2O + NH3 + NaOH → C6H4 (C (O) NH2) CO2Na + H2O
Il sale di sodio risultante dell’acido ftalamico viene decarbonilato tramite un riarrangiamento di Hofmann del gruppo ammidico, indotto dall’ipoclorito:
C6H4 (C (O) NH2) CO2Na + HOCl → C6H4NH2CO2H + NaCl + CO2
Un metodo correlato prevede il trattamento della ftalimmide con ipobromito di sodio in idrossido di sodio acquoso, seguito da neutralizzazione.
Nei tempi in cui la tintura indaco veniva ottenuta dalle piante, veniva degradata per dare acido antranilico.
Infatti l’acido antranilico è stato ottenuto per la prima volta dalla degradazione indotta da basi dell’indaco.
A livello biochimico l’acido antranilico viene biosintetizzato a partire dall’acido corismico. Negli organismi capaci della sintesi del triptofano, l’antranilato è un precursore dell’amminoacido triptofano, tramite l’attaccamento del fosforibosil pirofosfato al gruppo amminico.
A livello industriale, l’acido antranilico è un intermedio nella produzione di coloranti azoici e saccarina.
Esso e i suoi esteri vengono utilizzati nella preparazione di profumi che imitano il gelsomino e l’arancia, prodotti farmaceutici e prodotti che assorbono i raggi UV, nonché inibitori della corrosione dei metalli e inibitori della muffa nella salsa di soia.
Inoltre alcuni repellenti per insetti, a base di antranilato, sono stati proposti come sostituti del DEET (Dietiltoluamide).
L’Acido Antranilico è un aminoacido ed ha la funzione di coenzima nella produzione del latte materno. Esso quindi rappresenta un metabolita del Triptofano, altro importante Aminoacido cruciale per il benessere dell’apparato neuropsichico ed il suo mantenimento in salute.
Siccome esplica la funzione di coenzima nella produzione del latte materno, per questo è denominato, secondo una nomenclatura obsoleta delle vitamine, anche come vitamina L.
Infatti un tempo si pensava che l’acido antranilico fosse una vitamina e in quel contesto veniva chiamato vitamina L1, ma ora è noto che non è essenziale nella nutrizione umana.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.