Vanillina
Vanillina
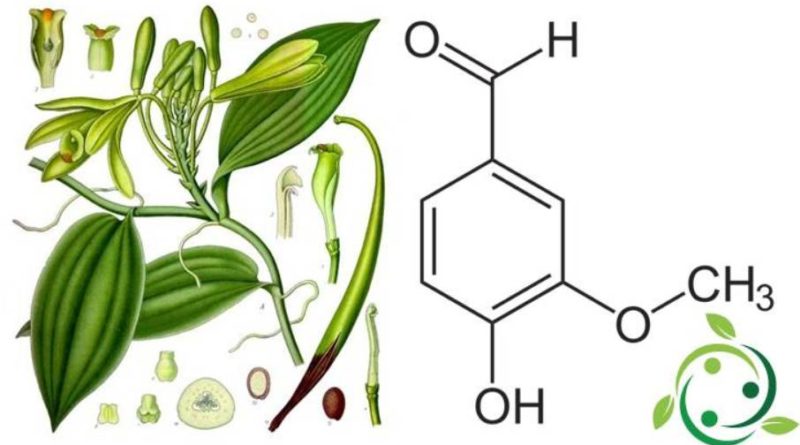
La vanillina, conosciuta anche come vaniglina, il cui termine nella nomenclatura ufficiale IUPAC è: 4-idrossi-3-metossibenzaldeide, ha formula bruta o molecolare:C8H8O3.
La vanillina è un’aldeide aromatica che a temperatura ambiente è un solido cristallino tendente al bianco, in base alla sua purezza, dal caratteristico aroma di vaniglia.
La vanillina è abbastanza solubile in acqua, molto più solubile in etanolo (500 g/l a 20 °C) e nei più comuni solventi organici.
La vanillina è una sostanza aromatica di provenienza naturale, ricavata dai frutti della vaniglia (Vanilla planifolia Jacks. ex Andrews, 1808), pianta che era coltivata nell’America Centrale già in epoca precolombiana.
La pianta venne scoperta dagli europei solo nei primi decenni del Cinquecento, con la conquista del Messico da parte di Hernán Cortés, quando videro che gli aztechi la usavano come un aroma per il cioccolato.
Bisogna arrivare però nel 1858 quando Nicolas-Theodore Gobley identificò la vanillina come sostanza a sé.
La prima sintesi di laboratorio si deve ad opera dei chimici tedeschi Ferdinand Tiemann e Wilhelm Haarmann che la ottennero producendola a partire dalla coniferina, presto sostituita dal più economico eugenolo, e poi dal guaiacolo.
Attualmente la vanillina sintetica viene impiegata più spesso dell’estratto di vaniglia naturale come aromatizzante in alimenti, bevande e prodotti farmaceutici.
La vanillina è contenuta nei semi della pianta di vaniglia in una percentuale che oscilla tra l’1,5% ed il 4%. L’estratto di vaniglia naturale contiene una miscela di diverse centinaia di composti oltre alla vanillina. Il prodotto sintetico si differenzia da quello di origine naturale per una maggiore probabilità di trovare deuterio oppure prozio tra il metossile ed il carbonile sul carbonio dell’anello.
L’aroma della vanillina viene ampiamente sfruttato dall’industria dolciaria (soprattutto nella produzione di gelati e cioccolato), cosmetica e farmaceutica (sia come eccipiente correttivo del sapore, sia come molecola base per la sintesi di altre sostanze).
Come molte altre sostanze di origine vegetale, la vanillina non si trova in natura come tale; piuttosto, è presente come aglicone all’interno di molecole glucosidiche.
Questo legame della vanillina con il glucosio, che impedisce tra l’altro la percezione del tipico aroma vanigliato, viene scisso alla morte del frutto per opera di enzimi idrolitici.
Tale reazione viene modulata dall’uomo attraverso l’iniziale macerazione dei baccelli in acqua calda, che ha lo scopo di arrestare i processi biochimici che avvengono nei tessuti vegetali viventi; dopodiché, per un paio di settimane, i frutti (baccelli appiattiti, lunghi 15-20 cm e larghi 8-10 mm) vengono fatti essiccare alla luce diurna, e avvolti in panni di lana durante la notte, dove vengono lasciati trasudare all’interno di scatole ermetiche.
In questa fase i legami glucosidici vengono scissi per idrolisi enzimatica, con liberazione di vanillina in forma libera; segue un processo finale di essicazione per arrestare i processi enzimatici, che a sua volta precede un periodo finale di invecchiamento più o meno lungo.
Questi processi produttivi tradizionali, piuttosto lenti, giustificano il costo nettamente più elevato (circa 100 volte) della vanillina naturale rispetto a quella sintetica, che attualmente copre la gran parte delle richieste commerciali.
I principali Paesi produttori di vanillina naturale sono ovviamente quelli in cui la vaniglia trova il clima ideale per la propria crescita: Messico, Madagascar, Indonesia, Cina, Tahiti e zone tropicali umide in genere.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.