Elenalina
Elenalina
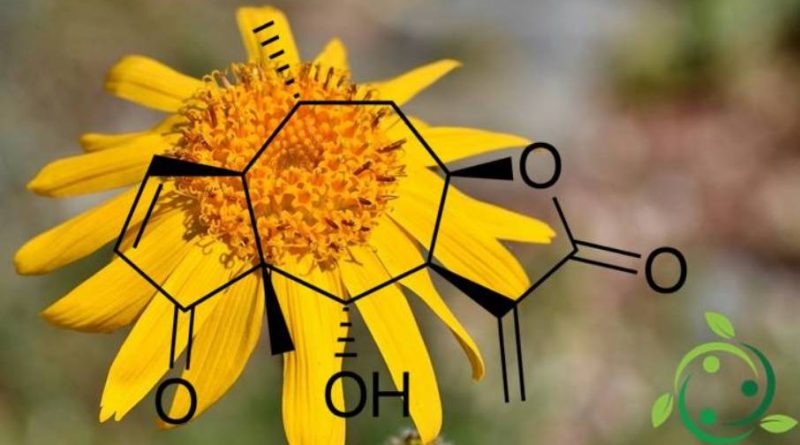
L’elenalina, il cui termine nella nomenclatura ufficiale IUPAC è: o (-) – 4-idrossi-4a, 8-dimetil-3,3a, 4a, 7a, 8,9,9a-octaidroazuleno [6,5-b] furan-2,5-dione, e la cui formula bruta molecolare è: C15H18O4 è un lattone sesquiterpenico che si trova in natura nelle piante di Arnica (Arnica montana L. e Arnica chamissonis var. foliosa (Nuttall) Maguire). L’elenalina è un composto altamente tossico e soprattutto i tessuti epatici e linfatici sono alquanto vulnerabili ai suoi effetti. L’ingestione di una quantità sufficiente di elenalina, tramite ingestione, produce gravi effetti gastroenterici ed emorragia interna del tratto digestivo.
Anche se tossica, l’elenalina possiede in vitro però alcuni effetti comprese le attività antinfiammatorie e antitumorali. L’elenalina può inibire alcuni enzimi, come la 5-lipossigenasi e il leucotriene C4 sintasi. Per questo motivo il composto o i suoi derivati possono trovare potenziali applicazioni mediche.
Al momento non esistono prove in vivo degli effetti antinfiammatori e antitumorali dell’elenalina.
In passato, estratti vegetali contenenti l’elenalina venivano usati come fitoterapia per vari trattamenti, come per la coagulazione del sangue, per la tensione muscolare e i disturbi reumatici.
Se applicata localmente sull’uomo, l’elenalina può causare dermatite da contatto in soggetti sensibili. Tuttavia, è considerata generalmente sicura quando applicata in tale modo.
La tossicità dell’elenalina è stata studiata in alcuni mammiferi come topi, ratti, conigli e pecore, dove la LD50 orale di elenalina è stata stabilita tra 85 e 150 mg / kg.
Studi su animali e in vitro hanno anche suggerito che l’elenalina può ridurre la crescita di Staphylococcus aureus e ridurre la gravità dell’infezione da S. aureus.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.