Santonina
Santonina
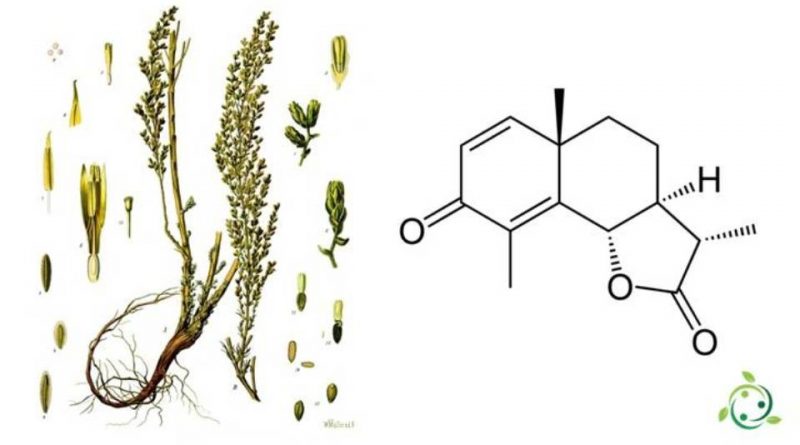
La santonina, il cui termine nella nomenclatura ufficiale IUPAC è: (3S, 3aS, 5aS, 9bS) -3,5a, 9-trimetil-3a, 5,5a, 9b-tetraidronafto [1,2-b] furan-2,8 (3H, 4H) –dione è un composto organico avente formula bruta C15H18O3.
La santonina appartiene alla serie dei composti sesquiterpenoidi.
Questo composto viene ricavato in natura ricavata dai capolini non espansi di Artemisia maritima var. Stechmanniana, mentre altri la fanno ricavare dal seme santo (Artemisia cina Berg & C.F. Schmidt ex Poljakov), pianta che era largamente usata in medicina per la sua azione di potente antielmintico, ma che ora è quasi completamente abbandonata a causa dei disturbi collaterali che può provocare (vomito, dolori addominali, ecc.).
La santonina paralizza i vermi parassiti (elminti), consentendo loro di essere espulsi dal corpo. La santonina ha l’effetto di paralizzare l’estremità anteriore (anteriore) del verme, mentre ha un effetto stimolante sull’estremità posteriore, a seconda della concentrazione. Per questo motivo, il verme non può coordinarsi e perde la sua capacità di mantenere la sua posizione nell’ospite.
Dal punto di vista chimico la santonina può essere convertita in acido santonico (C15H20O4 ) tramite idrolisi catalizzata a base seguita da un processo di riarrangiamento a più fasi.
La santonina è, inoltre, un composto otticamente levogiro .
Questo farmaco antielmintico si presenta allo stato solido come dei prismi piatti incolori, che diventano leggermente gialli per l’azione della luce e sono solubili in alcool, cloroformio e acqua bollente.
La santonina si dissolve in alcali con formazione di sali di acido carbossilico. La santonina, in soluzione di acido acetico, se esposta alla luce solare per circa un mese, viene convertita in acido fotosantonico (incolore) (C15H22O5) che è generalmente considerato meno tossico L’ estere etilico di quest’ultimo si ottiene quando una soluzione alcolica di santonina viene esposta alla luce solare (Sestini). Una colorazione gialla si sviluppa con l’esposizione alla luce della santonina.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.