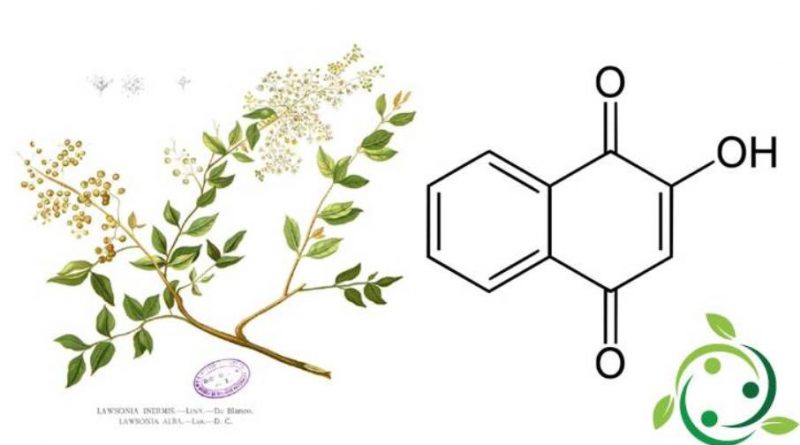
Lawsone
Lawsone
Il Lawsone, il cui termine nella nomenclatura ufficiale IUPAC è: 2-idrossi-1,4-naftochinone, conosciuto anche come acido hennotannico e la cui formula chimica bruta o molecolare è: C10H6O3 è un colorante rosso-arancio presente nelle foglie della pianta di henné (Lawsonia inermis L.) e nel fiore del giacinto d’acqua (Eichhornia crassipes (Mart.) Solms).
La procedura per l’estrazione del Lawsone dalla Lawsonia Inermis è complessa a causa della sua natura facilmente biodegradabile.
L’isolamento di questa molecola organica procede secondo quattro passaggi:
– estrazione con una solvente che generalmente è l’idrossido di sodio (NaOH);
– filtrazione su colonna mediante una resina di assorbimento macroporosa;
– trattamento con etanolo per rimuovere le impurità;
– fase finale di congelamento della sostanza per l’ottenimento di una polvere di colore giallo.
Il Lawsone è una molecola che viene utilizzata dagli esseri umani almeno da 5000 anni come colorante per i capelli.
Il Lawsone reagisce chimicamente con la cheratina presente nella pelle e nei capelli producendo una colorazione permanente che dura fino a quando la pelle o i capelli non ricrescono.
Il Lawsone, inoltre, assorbe fortemente la luce UV e gli estratti acquosi che contengono questa molecola possono avere un effetto abbronzante ed usati in creme solari molto efficaci.
Chimicamente, il lawsone è simile allo juglone, che si trova nelle noci.
Tra le altre possibili applicazioni, si ipotizza che il Lawsone sia soggetto ad una reazione simile alla sintesi di Strecker degli amminoacidi.
Di recente sono state condotte delle ricerche sulle potenziali applicazioni del lawsone nel campo della scienza forense.
Dal momento che il lawsone mostra molte somiglianze con il comportamento della Ninidrina, che è un indicatore altamente selettivo per il rilevamento degli amminoacidi, e che viene attualmente utilizzato quale reagente per rilevare le impronte digitali latenti, sono stati condotti delle ricerche per capire se il lawsone può essere usato in questo campo.
In questo campo, pur se la ricerca è in itinere, il Lawsone mostra risultati promettenti come reagente per il rilevamento delle impronte digitali a causa della sua fotoluminescenza massimizzata a 640 nm, che è abbastanza alta da evitare interferenze di fondo comuni alla ninidrina che invece reagisce con gli amminoacidi dando una colorazione azzurro-violetto (assorbimento a 570 nm), tranne con la prolina (è un amminoacido ciclico e presenta quindi un’ammina secondaria) con il quale forma un complesso giallo (assorbimento a 517 nm).
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.