Indacano
Indacano
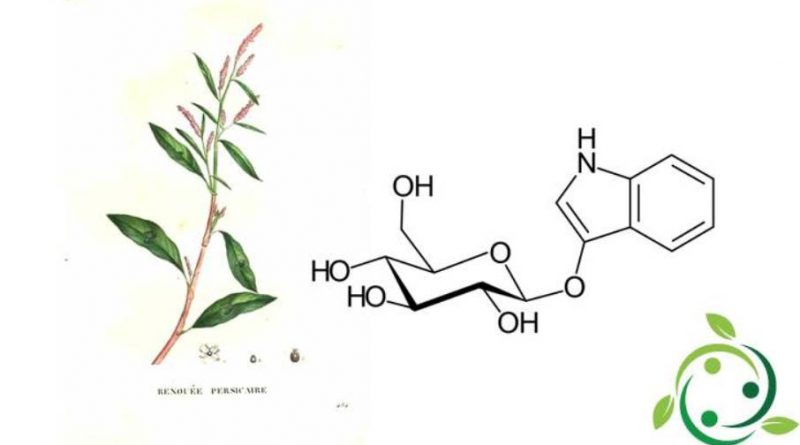
L’indacano o indicano, il cui termine nella nomenclatura ufficiale IUPAC è: (2R, 3S, 4S, 5R, 6S) -2- (idrossimetil) -6- (1H-indol-3-ilossi) tetraidropiran-3,4,5-triolo e la cui formula chimica molecolare è: C14H17NO6, è un glicoside presente nelle foglie di varie specie del genere indigofera e di altre piante (Isatis tinctoria, Persicaria tinctoria).
L’indacano è un composto organico incolore, solubile in acqua ed è un precursore della tintura indaco. Questa tintura, soprattutto un tempo, prima dell’avvento della chimica di sintesi dava appunto il colore indaco che è un colore intenso e saturo, tradizionalmente considerato un colore nello spettro visibile, nonché uno dei sette colori dell’arcobaleno: il colore tra viola e blu.
L’indacano passa nell’acqua di macerazione delle foglie stesse, e per idrolisi (provocata da enzimi anch’essi presenti) si scinde in β-D-glucosio e indossile.
L’indacano, a livello umano, è presente nel sangue in piccola quantità (indacanemia) ed è eliminato per via urinaria (indacanuria).
In alcuni stati morbosi indacanemia e conseguentemente l’indacanuria aumentano considerevolmente.
Questo è dovuto ad un difetto nel metabolismo del triptofano. Il triptofano viene prima convertito in indolo, quindi in indacano dai batteri nell’intestino. L’indicano viene quindi escreto nelle urine e da lì dopo l’esposizione all’aria, viene convertito in colorante blu indaco a causa dell’ossidazione dell’ossigeno atmosferico.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.