Serina
Serina
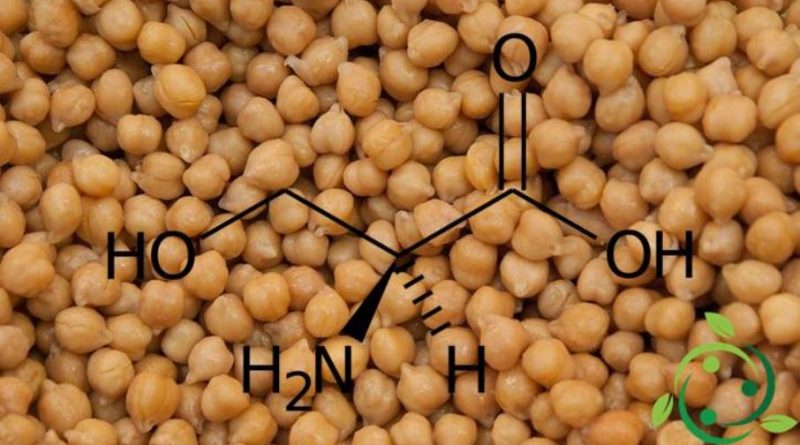
La serina, il cui termine nella nomenclatura ufficiale IUPAC è: acido 2(S)-ammino-3-idrossipropanoico, è un amminoacido polare con formula bruta o molecolare: C3H7NO3.
La molecola della serina è chirale e l’enantiomero L è uno dei 20 amminoacidi ordinari, con un gruppo laterale legato ad un gruppo idrossile.
La serina non è un amminoacido essenziale, in quanto l’organismo umano è in grado di sintetizzarlo e che si trova coinvolto in alcuni difetti ed effetti gravi del diabete.
Infatti il suo gruppo laterale può subire reazione di O-glicosilazione, cioè l’addizione di una molecola di glucosio; questo la coinvolge nella catena di reazioni biochimiche che spiegano alcuni gravi effetti del diabete.
È un aminoacido che deriva in parte dall’amminoacido glicina ed in parte può essere assunto attraverso l’alimentazione. La serina, attraverso una reazione catalizzata dalla serina metiltransferasi, SHMT, si trasforma in glicina trasferendo un atomo di carbonio al tetraidrofolato che si trasforma in metilentetraidrofolato la forma più versatile dei “folati” nella cellula.
La sua principale funzione è quella di promuovere la sintesi della creatina. Ha inoltre la funzione di produrre energia cellulare, in modo da poter apportare energia e riserve caloriche al fegato e al muscolo. Tra gli alimenti maggiormente ricchi di serina ricordiamo: pasta all’ uovo, ceci, fave, lenticchie, arachidi, nocciole, coniglio, pollo, pesce, formaggio, e uovo di gallina.
La serina (come la tirosina e la treonina) può anche subire a livello della sua catena laterale una reazione di fosforilazione con la formazione di fosfoserina.
La serina può essere destinata alla produzione di energia metabolica, sotto forma di ATP. Per farlo deve essere prima deidratata in una reazione catalizzata dall’enzima serina deidratasi, che rimuove una molecola d’acqua sotto forma di un gruppo idrossilico e di un atomo di idrogeno dal Cα. L’intermedio che si forma subisce una reazione di transamminazione: cede il suo gruppo amminico all’α chetoglutarato e si trasforma in piruvato.
A sua volta il piruvato può essere decarbossilato ossidativamente in acetilCOA dal complesso della piruvato deidrogenasi, oppure può essere carbossilato in ossalacetato da parte dell’enzima piruvato carbossilasi (prima tappa della gluconeogenesi) e trasformarsi in glucosio.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.