Norbaeocistina
Norbaeocistina
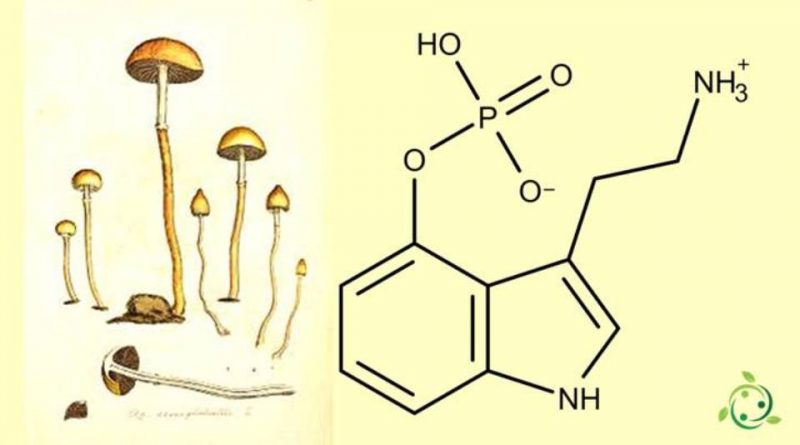
La norbaeocistina il cui termine nella nomenclatura ufficiale IUPAC è: 3-[2-(ammonioetil)]-1H-indol-4-il idrogenofosfato è un alcaloide.
Questa sostanza ha formula bruta o molecolare: C10H13N2O4P.
Dal punto di vista chimico la norbaeocistina è una triptamina dalla struttura chimica correlata a quella della psilocibina e in modo ancora più marcato della baeocistina. A differenza di quest’ultima non è sicuro che la molecola contribuisca agli effetti enteogenici della psilocina.
La norbaeocistina è presente in natura, in dosi apprezzabili, in un fungo del genere Psilocybe (Psilocybe baeocystis Singer & A.H.Smith (1958)).
Il Psilocybe baeocystis è un fungo della famiglia delle Hymenogastraceae che contiene i composti allucinogeni psilocibina, psilocina e baeocistina.
La norbaeocistina è un derivato N-demetilato della baeocistina (a sua volta un derivato N-demetilato della psilocibina) e un derivato fosforilato della 4-idrossitriptamina. La 4-idrossitriptamina è nota per essere un isomero di posizione della serotonina, che è la 5-idrossitriptamina.
Recentemente, alcuni ricercatori hanno dimostrato che la norbaeocistina potrebbe essere un intermedio nella biosintesi della psilocibina.5 Test in vitro hanno dimostrato che l’L-triptofano viene convertito in triptamina dall’enzima PsiD che viene poi convertito in 4-idrossitriptamina dall’enzima PsiH. PsiD può anche sintetizzare la 4-idrossitriptamina direttamente dal substrato 4-idrossi-L-triptofano. La norbaeocistina viene quindi sintetizzata dall’enzima PsiK. I ricercatori propongono che ulteriori gruppi metilici vengano aggiunti dall’enzima PsiM, generando baeocistina (una metilazione) e psilocibina (due metilazioni).
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.