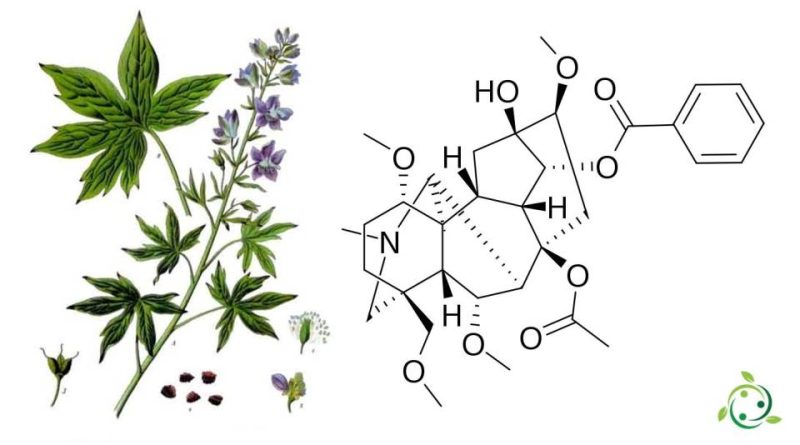
Delphinine
Delphinine
Delphinine, whose term in the official IUPAC nomenclature is: 8- (acetyloxy) -13-hydroxy-1,6,16-trimethoxy-4- (methoxymethyl) -20-methylaconitan-14-benzoate is a toxic diterpenoid alkaloid.
This substance has a brute or molecular formula: C33H45NO9 and is found in plants of the genera Delphinium and Atragene of the Ranunculaceae family.
Delphinine is the main alkaloid present in the seeds of Stavesacre (Staphisagria macrosperma Spach, 1839) which was used, especially in the past, for a well-known herbal treatment for body lice.
Delphinine is also chemically similar to aconitine, acting as an allosteric modulator of sodium channels and inducing low blood pressure, slow heart rate and abnormal heart rhythms. These effects make it highly poisonous (LD50 1.5-3.0 mg / kg in rabbits and dogs; frogs are about 10 times more sensitive).
Although it has been used in some alternative medicines (such as herbal medicine), most of the medical community discourages its use due to its extreme toxicity.
The dolphin was isolated from the Staphisagria macrosperma by the French chemists Lassaigne and Feneulle, in 1819.
Although the discovery of the dolphin dates back over 200 years, its molecular structure was established in its currently accepted form only in the early 1970s. At that time, Wiesner’s research group corrected the stereochemistry of the methoxy group in C-1 from the – configuration to the α- configuration. Therefore, any drawing of the delphinine molecule that appears prior to 1971-1972 is likely to show incorrect stereochemistry in C-1.
From a pharmacological point of view, following its discovery and isolation in its crystalline form, the properties of delphinine were extensively studied in the 19th century, despite its molecular structure being unknown. Some of these investigations are likely to have been carried out with impure drugs and should be interpreted with caution. More recent studies have focused on the cardiovascular toxicity of delphinine.
In general, the pharmacology of delphinine appears to resemble that of aconitine, although the acute toxicity of delphinine appears to be lower than that of aconitine in experimental animals.
Warning: The information shown is not medical advice and may not be accurate. The contents are for illustrative purposes only and do not replace medical advice.