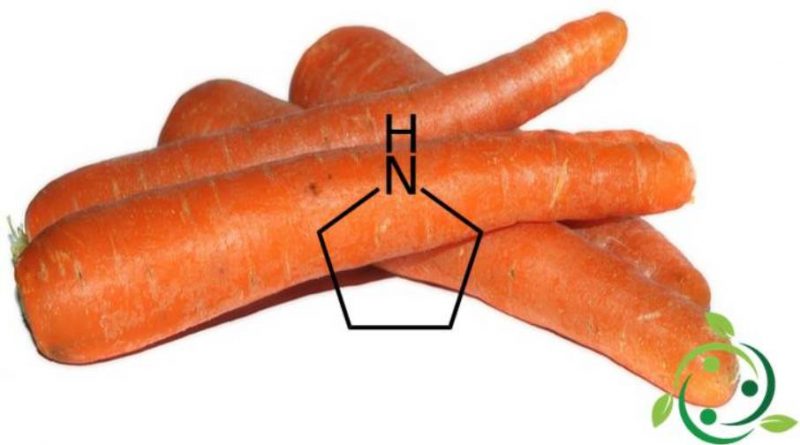
Pirrolidine
Pirrolidine
Pyrrolidine or tetrahydropyrrole, is an organic compound with brute C4H9N molecular formula. It is a cyclic secondary amine consisting of a five-membered heterocyclic ring, of which 4 are carbon and 1 is nitrogen.
Pyrrolidine is a very basic basis. Its conjugated acid possesses pKa = 11.27.
Pyrrolidine is a molecule which at room temperature is in the form of a colorless or yellow liquid; it has a characteristic pungent ammonia and fish smell.
Pyrrolidine is present in nature and is found in the leaves of some plants; among these we remember tobacco, carrot and belladonna.
The heterocyclic ring of pyrrolidine is found in many natural alkaloids, such as nicotine and hygrine. In addition to many drugs, it becomes part of the structure of racetams and of various pesticides. Furthermore, the central structure of the proline and hydroxyproline amino acids is pyrrolidine.
In addition to nature it can be obtained by chemical synthesis. It can be obtained by reacting tetrahydrofuran with ammonia in the gaseous phase, in the presence of an aluminum oxide catalyst. Pyrrolidine can also be obtained by hydrogenation of pyrrole, which can be obtained in turn by dehydrogenation of pyrrole, by reverse reaction.