Tirosina
Tirosina
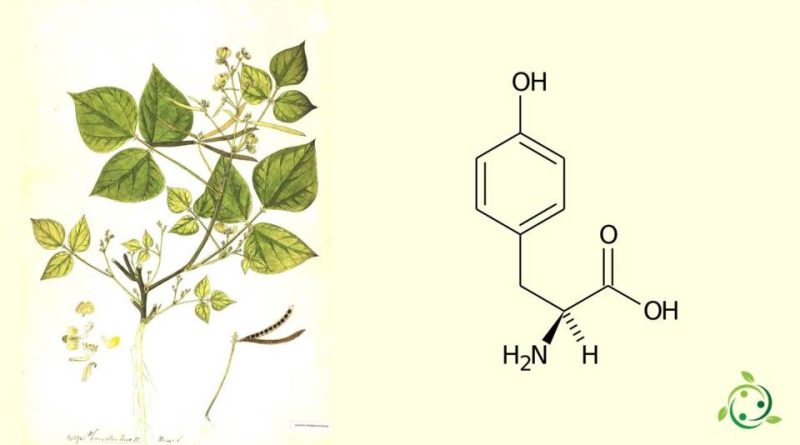
La tirosina, il cui termine nella nomenclatura ufficiale IUPAC è: acido 2(S)-ammino-3-(4-idrossifenil)propanoico è un amminoacido polare con molecola chirale.
La tirosina viene conosciuta anche con le abbreviazioni: Y e TYR ed ha formula bruta o molecolare: C9H11NO3.
La tirosina è stata scoperta dal chimico tedesco Justus von Liebig all’interno della caseina, ed il nome tirosina viene greco tyros, che significa formaggio.
L’enantiomero L è uno dei 20 amminoacidi ordinari e il suo gruppo laterale è un p-idrossibenzile.
La tirosina è un amminoacido non essenziale per l’organismo umano in quanto è in grado di sintetizzarlo.
La tirosina è biologicamente importante in quanto è un precursore di vari ormoni, quali la tiroxina (un ormone tiroideo) e le catecolammine (dopamina, noradrenalina e adrenalina), e della melanina.
Tirosina negli alimenti –
Tra i cibi apportatori di tirosina ricordiamo i prodotti della soia, il pesce, il pollo, il tacchino, le mandorle, le banane, il latte ed i suoi derivati, i semi si sesamo, l’avocado ed i semi di zucca.
Biosintesi –
La tirosina viene prodotta nelle piante e nella maggior parte dei microrganismi a partire dal prefenato, intermedio della via dell’acido scichimico. Il prefenato è sottoposto a decarbossilazione ossidativa NAD-dipendente con ritensione dell’idrossile, dando luogo al p-idrossifenilpiruvato, successivamente sottoposto ad una reazione di transaminazione dove il glutammato è il donatore del gruppo amminico.
I mammiferi hanno la capacità di sintetizzare la tirosina dall’aminoacido essenziale fenilalanina (phe), introdotto tramite l’alimentazione. La conversione della fenilalanina in tirosina è catalizzata dall’enzima fenilalanina idrossilasi, un’ossigenasi a funzione mista che utilizza un cofattore pterinico, la tetraidrobiopterina. Questo enzima catalizza la reazione di conversione aggiungendo un gruppo ossidrile sull’atomo di carbonio in posizione 6 dell’anello aromatico della fenilalanina.
Proprietà –
La tirosina è un aminoacido prezioso per il corretto funzionamento del nostro cervello. Regola l’umore e la risposta allo stress. È il precursore di importanti neurotrasmettitori che vanno a regolare l’umore e le capacità cognitive, quali dopamina, adrenalina e noradrenalina.
Per questo motivo viene impiegata nella cura della depressione, dei disturbi del sonno e dei deficit dell’attenzione. Inoltre migliora la risposta di adattamento del corpo allo stress, tanto che veniva impiegata dai piloti dei cacciabombardieri.
Trova impiego anche nell’uso sportivo.
La tirosina è un amminoacido precursore delle catecolammine (adrenalina, noradrenalina e dopamina) nel cervello, ed è stato rilevato che la sua supplementazione possa accelerare la sintesi di queste molecole nel sistema simpatoadrenale. Gli studi sugli effetti della supplementazione di tirosina in ambito sportivo non hanno registrato effetti rilevanti sulla prestazione anche ad alti dosaggi. La tirosina non ha dimostrato alcun miglioramento della performance anche se combinato con i carboidrati.
Alcune evidenze empiriche suggeriscono che la tirosina possa avere un impatto come stimolante se assunta come supplemento nel pre-allenamento in concomitanza con altre sostanze, esercitando un effetto sinergico. Studi su animali hanno dimostrato che la tirosina possa potenziare gli effetti dell’associazione caffeina/efedrina.
Carenze di tirosina –
Le carenze di tirosina sono piuttosto rare ed associate perlopiù a casi malnutrizione calorico-proteica. Bassi livelli di questo amminoacido sono stati associati a ipotensione, bassa temperatura corporea e ad ipotiroidismo. Tirosina – Stress – Depressione. Deficit di tirosina possono registrarsi negli individui fenilchetonuretici, a causa della necessità di allontanare nel modo più assoluto tutte le fonti alimentari di fenilalanina dalla propria dieta. Il deficit viene compensato inserendo l’amminoacido nei supplementi amminoacidici che questi individui sono costretti ad assumere quotidianamente in quantità importanti.
Eccessi di tirosina –
L’eccessiva assunzione di tirosina è stata messa in relazione con lesioni oculari, ritardi della crescita e cali di appetito. Tuttavia, la tirosina è generalmente ben tollerata e solo una piccola percentuale di individui ha lamentato problemi di nausea, mal di testa, affaticamento, bruciori di stomaco e dolori articolari dopo la sua assunzione.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.