α-terpineolo
α-terpineolo
L’ α-terpineolo, il cui termine nella nomenclatura ufficiale IPAC è: 2-(4-Metil-1-cicloex-3-enil)propan-2-olo è un alcol naturale monoterpenico con formula bruta o molecolare: C10H18O.
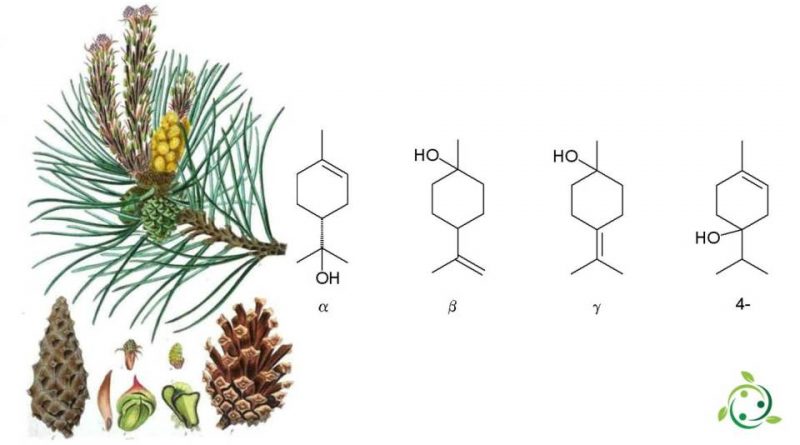
L’α-terpineolo è un composto isolato da una varietà di fonti come l’olio essenziale di pino e l’olio petitgrain. La sua presenza è in genere accompagnata da piccole percentuali dei due isomeri β-terpineolo e γ-terpineolo. La miscela naturale dei tre isomeri prendi il nome generico di terpineolo.
I terpenoidi sono terpeni modificati dall’aggiunta di un gruppo funzionale, che in questo caso è un alcol. Esistono quattro isomeri: α-, β-, γ-terpineolo e terpinen-4-olo. Il terpineolo è solitamente una miscela di questi isomeri con α-terpineolo come costituente principale.
Il terpineolo ha un odore gradevole simile al lillà ed è un ingrediente comune in profumi, cosmetici e aromi.
Sebbene questa sostanza sia presente in natura, è comunemente prodotto dall’alfa-pinene più facilmente disponibile. Un percorso alternativo di sintesi è quella che parte dal limonene.
Nella sintesi a partire dal limonene, quest’ultimo reagisce con l’acido trifluoroacetico, secondo la regola di Markovnikov, arrivando a un intermedio trifluoroacetato, che è facilmente idrolizzato con idrossido di sodio ad α-terpineolo con selettività del 7%. I prodotti collaterali sono il β-terpineolo in una miscela dell’isomero cis, dell’isomero trans e del 4-terpineolo.
La biosintesi dell’α-terpineolo procede invece dal geranil pirofosfato, che rilascia pirofosfato per dare il catione terpinile. Questo carbocatione è il precursore di molti terpeni e terpenoidi. La sua idrolisi dà terpineolo.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.