Acido 4-idrossibenzoico
Acido 4-idrossibenzoico
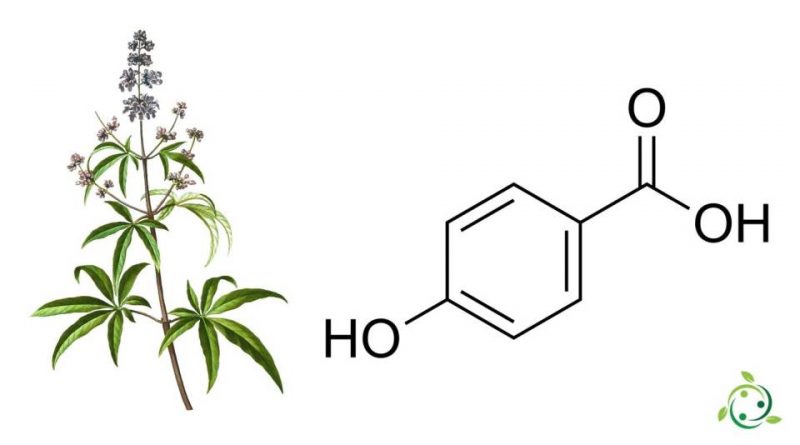
L’acido 4-idrossibenzoico, conosciuto con le abbreviazioni di 4-HBA o PHBA o con i nomi alternativi di acido para idrossibenzoico o acido p-idrossibenzoico è un acido organico con formula bruta o molecolare: C7H6O3.
Si tratta di un acido monoidrossibenzoico la cui struttura è costituita da un anello benzenico con un gruppo carbossilico (-COOH), come nell’acido benzoico, e un gruppo idrossilico (-OH) come sostituente in posizione 4 dell’anello benzenico.
L’acido 4-idrossibenzoico, dal punto di vista fisico, è un solido cristallino bianco leggermente solubile in acqua e cloroformio ma più solubile in solventi organici polari come alcol, etere e acetone.
Questa sostanza ha una nota aromatica di nocciola e fenolo; dal JECFA e dal regolamento UE è classificato come aroma in ambito alimentare.
L’acido 4-idrossibenzoico o suoi esteri e glucosidi, sono presenti in molte specie viventi appartenenti a famiglie e generi diversi: dai batteri alle alghe, dai funghi all’uomo. Negli esseri umani, l’acido 4-idrossibenzoico è coinvolto nella biosintesi dell’ubichinone.
Acido 4-idrossibenzoico è ampiamente utilizzato nella sintesi organica, in particolare i suoi esteri, compresi metil 4-idrossibenzoato (paraben Kap), etil (etil paraben), propile, butile, isopropile, isobutile, sono utilizzati come additivi alimentari, nella salsa di soia, aceto, bevande, aromi di frutta, frutta e verdure, sottaceti, ecc; inoltre è anche ampiamente utilizzato nei prodotti alimentari, cosmetici, farmaceutici anti-corrosione, muffa e fungicidi, ecc.
L’acido 4-idrossibenzoico, come altri parabeni, viene utilizzato anche come base per i coloranti, antiparassitari e prodotti intermedi.
L’acido 4-idrossibenzoico trova impiego nell’industria dei conservanti e come fungicida. È in grado di inibire la crescita di muffe ed è un ottimo conservante.
L’acido 4-idrossibenzoico è una sostanza presente in natura ed isolato da radici e parti aeree di molte piante; ad esempio: Vitex agnus-castus, Vitex negundo, Vitis vinifera, Hypericum perforatum, Daucus carota, Elaeis guineensis, Euterpe oleracea, Fagara macrophylla, Xanthophyllum rubescens, Paratecoma peroba, Tabebuia impetiginosa, Pterocarpus santalinus, Catalpa bignonioides, Areca catechu, Roystonea regia, Mespilus germanica, Arabidopsis thaliana, Phyllanthus acidus, Macrotyloma uniflorum.
Inoltre lo si riscontra anche in varie alghe come: Spongiochloris spongiosa , Anacystis nidulans, Sargassum tenerrimum, Sargassum ilicifolium, Sargassum cinereum.
Si suppone, inoltre, che sia uno dei componenti attivi di un fungo che ha avuto per lungo tempo un utilizzo medicinale: Ganoderma lucidum.
È stato isolato anche in molti funghi di vari generi: Agaricus, Boletus, Cantharellus, Clitocybe, Laccaria, Lactarius, Pleurotus, Russula, Suillus, Termitomyces, ecc..
Inoltre il metabolismo di vari batteri comporta la produzione di 4-HBA: Cryptanaerobacter phenolicus , Clostridium hydroxybenzoicum, Sporotomaculum hydroxybenzoicum.
La grande diffusione in natura dell’acido 4-idrossibenzoico, oltre che al massivo utilizzo di suoi esteri nella cosmesi, hanno prodotto la necessità di definirne i livelli di esposizione nell’uomo.
L’utilizzo massivo dell’acido 4-idrossibenzoico nella produzione di conservanti cosmetici o di additivi alimentari è in parte dovuto alla sua tossicità acuta molto bassa: LD 50 è 2200 mg / kg nei topi (orale).
Come precursore, ma anche come metabolita, dei parabeni è stato oggetto di indagine per una sospetta attività estrogenica. Uno studio valido e affidabile con somministrazione ripetuta di dosaggi orali fino a 1000 mg / kg di peso corporeo utilizzando ratti (42 giorni) e studi validi e affidabili su ratti e topi femmine immaturi con somministrazione ripetuta di dosaggi fino a 100 mg / kg di peso corporeo per 3 giorni consecutivi (saggi uterotrofici) erano disponibili e principalmente utilizzati per la valutazione.
Lo studio di tossicità a dose ripetuta nei ratti (Nagao et al., 1997) non ha indicato alcun effetto sugli organi riproduttivi di ratti femmine e maschi o / e aspetti funzionali della fertilità da parte dell’acido p-idrossibenzoico. I test uterotrofici affidabili (Twomey 2000b, Hossaini et al. 2000) sono risultati negativi per l’acido p-idrossenzoico in ratti e topi in concentrazioni fino a 100 mg / kg di peso corporeo.
In sintesi, esiste un numero sufficiente di studi sufficientemente affidabili e validi per la valutazione dei possibili effetti estrogenici dell’acido p-idrossibenzoico. Ad oggi nessuno di questi studi ha mostrato un effetto estrogenico dell’acido p-idrossibenzoico.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.