Acido Elaidinico
Acido Elaidinico
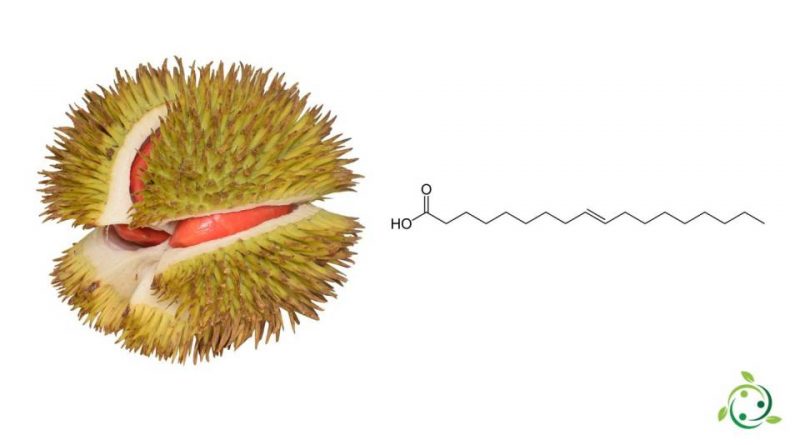
L’acido elaidinico o acido elaidico o, ancora, acido trans-oleico ed il cui termine nella nomenclatura ufficiale IUPAC è: acido trans-9-ottadecenoico è un acido grasso presente in natura.
L’acido elaidinico ha formula bruta o molecolare: C18H34O2.
Questo composto è un acido grasso a 18 atomi di carbonio monoinsaturo trans in posizione 9. Si tratta dell’isomero dell’acido oleico.
L’acido elaidinico è presente in natura, come altri acidi grassi trans o coniugati, e si forma per il processo di bioidrogenazione dovuto a batteri, normalmente gram negativi, in ambiente anaerobico.
Lo si ritrova anche nell’olio di sansa per via dell’isomerizzazione dell’acido oleico durante l’essiccamento delle sanse in contenitori metallici che catalizzano la reazione.
L’acido elaidinico si trova naturalmente in piccole quantità anche nel latte caprino e bovino (circa lo 0,1% degli acidi grassi) e in alcune carni. Presnte per circa il 2,50% dei grassi del frutto della specie durian (Durio graveolens Becc., 1889).
Tra le proprietà dell’acido elaidinico si ricorda che aumenta l’attività della proteina di trasferimento del colesterolo plasmatico (CETP) che abbassa il colesterolo HDL.
L’acido elaidinico fu individuato per la prima volta dal farmacista francese, Poutet JJE, che gli attribuì il nomedi: acide élaidique da Boudet, che chiamò elaidine, il prodotto di idrogenazione delle trioleine.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.