Guttaperca
Guttaperca
La guttaperca è una macromolecola di origine vegetale molto simile alla gomma naturale o caucciù, da cui differisce però per molte proprietà, tra cui l’assenza di elasticità.
Il termine guttaperca deriva dall’inglese gutta-percha, a sua volta originato dal malese gĕtah pĕrcah che significa “gomma di perca”, dal nome dell’albero da cui è ricavata.
La guttaperca è il lattice disseccato di varie specie di alberi della famiglia delle Sapotacee, indigene della regione indomalese.
La specie dalla quale dapprima venne ricavata la guttaperca fu il Palaquium gutta Burck ma poiché si estraeva il lattice abbattendo gli alberi, anziché incidendoli, la specie fu quasi distrutta; oggi la guttaperca si ottiene principalmente dai P. oblongifolium Burck, P. borneense Burck, P. Treubii Burck e dalla Payena Leerii Benth. e Hook.
La zona in cui queste piante crescono è notevolmente più ristretta di quella delle piante da caucciù. La coltivazione in piantagione è limitatissima (piantagioni Tijpetir a Giava e Selborue a Sumatra).
La guttaperca è usata in oriente da epoca remota. In Europa fu importata verso la metà del sec. XVII da Tradescant, ma non ebbe applicazione. Solo nel 1847 la ditta Siemens ne iniziò l’utilizzazione per la preparazione di cavi sottomarini, sfruttandone l’alto potere isolante e la proprietà di essere impermeabile all’acqua e relativamente elastica.
Il suo impiego nell’industria si è ridotto però nel corso del XX secolo, sostituita per lo più da resine sintetiche.
Viene impiegata soprattutto per fogli, tubi, abiti impermeabilizzati e altri oggetti; in particolare si usa in odontoiatria per chiudere i canali dopo la devitalizzazione della polpa, la parte più interna del dente.
La guttaperca fu introdotta in Italia per la prima volta nei primi del Novecento da Giovanni Battista Pirelli, fondatore dell’omonima ditta.
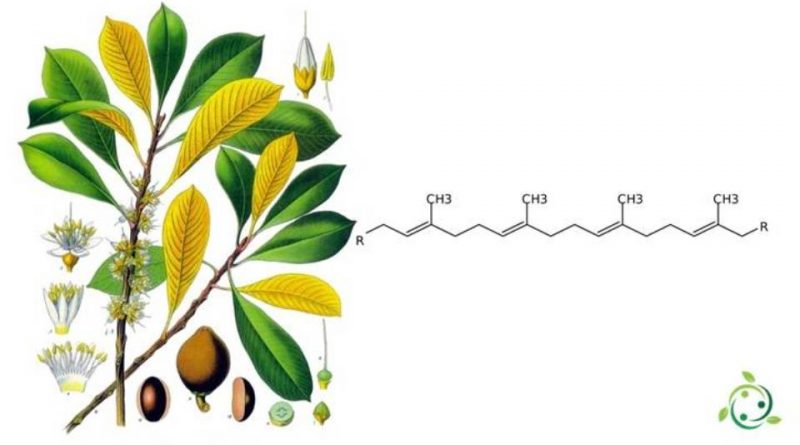
Dal punto di vista chimico la guttaperca è un politerpene, ovvero una macromolecola appartenente alla famiglia dei terpeni. In altre parole è un polimero dell’isoprene con più di 40 atomi di carbonio. È formato per addizione 1,4-trans, ed il risultato è una materia plastica elastomerica che ha diversi impieghi industriali.
Inoltre, dal punto di vista della costituzione la guttaperca e il caucciù sono uguali. In ambedue i polimeri infatti la successione degli atomi e dei legami nell’unità monomerica è identica. La presenza del doppio legame però determina un fenomeno di isomeria sterica, quindi a seconda di come si dispone il monomero di base, abbiamo due possibilità:
– Configurazione trans, che produce una molecola di guttaperca;
– Configurazione cis, che produce una molecola di caucciù.
Questa stereoisomeria, all’interno del polimero, ha un effetto notevolissimo sulle proprietà dello stesso.
Così mentre il caucciù risulta essere un elastomero amorfo, la guttaperca a temperatura ambiente è un polimero semicristallino. Dato dal fatto che la configurazione trans, meno ingombrante, favorisce l’impaccamento.
Dal punto di vista fisico il caucciù presenta una temperatura di transizione vetrosa (Tg) di −70 °C mentre la guttaperca ha una Tg di -38 °C e fonde a 60 °C.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.