Isoleucina
Isoleucina
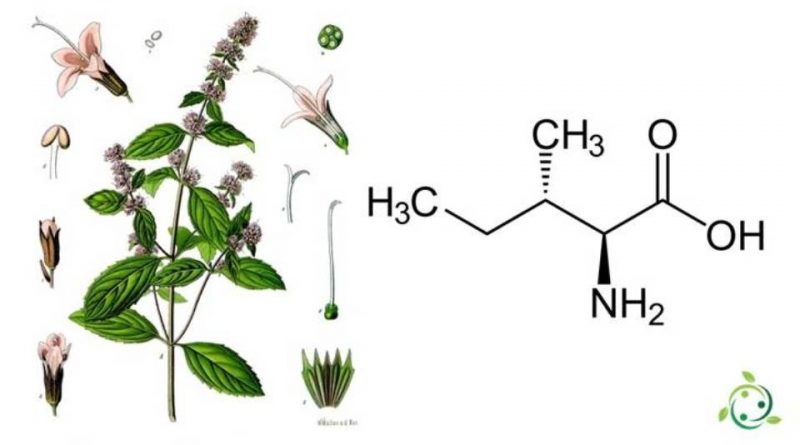
L’ Isoleucina, il cui termine nella nomenclatura ufficiale IUPAC è L-isoleucina (conosciuta anche con l’abbreviazione Ile o I), o acido (2S,3S)-2-ammino-3-metilpentanoico, è un α-amminoacido.
L’ Isoleucina fu scoperta nel 1903 dal chimico tedesco Felix Ehrlich, che la individuò nella melassa della barbabietola.
L’isoleucina sintetica fu segnalata invece per la prima volta nel 1905 dal chimico francese Louis Bouveault.
L’ Isoleucina ha formula bruta o molecolare: C6H13NO2 con molecola è chirale che contiene un α-ammino gruppo, un gruppo carbossilico e una catena laterale alifatica, un sec-butile, classificata come apolare e non carica (a pH fisiologico).
L’enantiomero L è uno dei 20 amminoacidi ordinari e si riferisce allo stereoisomero acido-(2S,3S)-2-amino-3-metilpentanoico: questa è la configurazione naturale.
Dal punto di vista biochimico l’isoleucina fa parte, assieme alla treonina, della ristretta famiglia di α-amminoacidi naturali contenenti due centri stereogenici. I possibili stereoisomeri sono quattro e posseggono rispettivamente configurazione assoluta: (2S,3S), (2R,3R), (2S,3R) e (2R,3S).
Quando si parla di L-treonina ci si riferisce allo stereoisomero (2S,3R), il più rappresentato in natura.
L’ Isoleucina è un amminoacido essenziale per gli esseri umani in quanto il corpo non può sintetizzarlo e deve quindi essere assunto con la dieta.
L’isoleucina viene invece sintetizzata in altri organismi, come i batteri, a partire dal piruvato utilizzando enzimi biosintetici della leucina. Nella trascrizione genetica è codificata nei codoni ATT, ATC, ATA.
Essendo un nutriente essenziale, non sintetizzato dal corpo umano, l’isoleucina deve essere assunta con la dieta, solitamente come componente di proteine.
Nelle piante e nei microrganismi è sintetizzata in vari passaggi, partendo da acido piruvico e alpha-chetoglutarato.
Le fonti nutrizionali di questo amminoacido, sebbene non sia sintetizzato dagli animali, sono soprattutto quelle degli alimenti di origine animale. Gli alimenti con più alto contenuto di isoleucina sono: uova, proteine della soia, alga marina, tacchino, pollo, agnello, formaggio e pesce. Tuttavia l‘isoleucina è presente anche in alcune piante come nello Scalogno (Allium ascalonicum L.), nella menta piperita (Mentha × piperita) ed altre ancora.
Il fabbisogno giornaliero, che viene raccomandato dall’Organizzazione mondiale della sanità per questo amminoacido essenziale, è pari 20 mg/kg di peso corporeo. Ad esempio, il valore raccomandato per un individuo di 70 kg è 1400 mg.
Dal punto di vista biochimico l’isoleucina è un amminoacido sia glucogenico sia chetogenico.
Successivamente alla transaminazione con alpha-ketoglutarato la catena laterale alifatica può essere convertita sia in succinil-coenzima A, e inserita nel ciclo dell’acido citrico per l’ossidazione, o convertita in acido ossalacetico per la gluconeogenesi (perciò glucogenico).
L’isoleucina può anche essere convertita in acetil-coenzima A e inserita nel ciclo dell’acido citrico per condensazione con acido ossalacetico per formare citrato.
Nei mammiferi, comunque, l’acetil-coenzima A non può essere riconvertito a carboidrato ma può essere impiegato nella sintesi di corpi chetonici o acidi grassi, motivo per cui l’isoleucina è un amminoacido chetogenico.
L’incapacità di metabolizzare l’isoleucina, insieme a altri amminoacidi, è associata con la malattia MSUD che consiste nella decolorazione delle urine del paziente e nell’odore caratteristico di queste, da cui deriva il nome della patologia e che, nei casi più gravi può condurre a danni alle cellule nervose e al decesso.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.