Acidi uronici
Acidi uronici
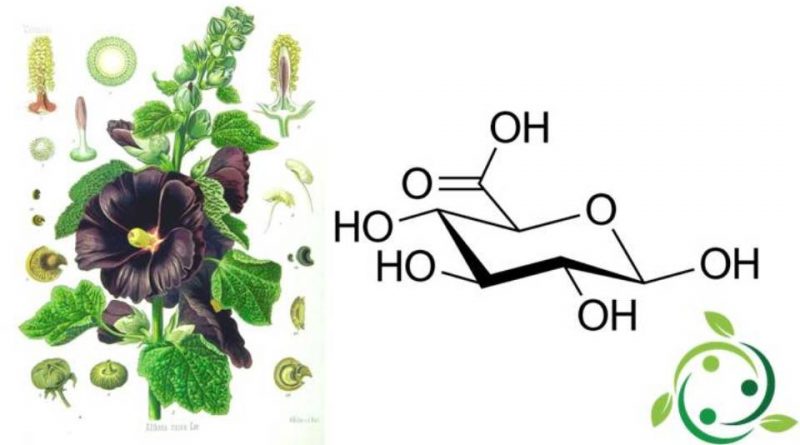
Gli acidi uronici sono degli acidi monocarbossilici derivati dai monosaccaridi aldosi o chetosi, ottenuti per ossidazione di un gruppo alcolico primario di un chetosio o di un aldosio, generalmente a 5 o 6 atomi di carbonio.
Gli acidi uronici prendono il nome dei corrispondenti monosaccaridi.
Si tratta di acidi alquanto diffusi in natura: ricordiamo l’acido glucuronico che si ritrova in molti polisaccaridi presenti nel mondo animale e vegetale;, l’acido galatturonico che si rinviene nelle pectine; l’acido mannuronico che è il componente principale dell’acido alginico ecc.
Gli acidi uronici sono derivati principalmente attraverso processi di biosintesi in cui intervengono appositi enzimi. Per diventare acidi alduronici, gli zuccheri aldosi subiscono un’ossidazione enzimatica altamente specifica, che risparmia il gruppo carbonilico posto sul C1 ma agisce solamente sull’ossidrile del C6, posto all’estremità della catena carboidratica: dunque, un acido alduronico presenta ancora il vecchio gruppo aldeidico sul C1 ed un nuovo gruppo carbossilico sul C6.
Ovviamente, al pari di un aldoso, un acido uronico esiste soprattutto in forma ciclica, con il C1 ed il C5 impegnati nel legame emiacetalico tipico di tutti i carboidrati piranosici e furanosici.
Gli acidi uronici svolgono importanti funzioni biochimiche; si ricorda l’acido glucuronico, derivante dal D-glucosio, che permette l’escrezione delle sostanze tossiche attraverso l’urina per coniugazione degli alcoli e fenoli esogeni (come ad esempio l’anestetico propofol) a livello degli epatociti con formazione di un legame glicosidico che produce una molecola idrosolubile. Oppure l’acido iduronico che è il componente di alcuni complessi strutturali come i proteoglicani.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.