Tujone
Tujone
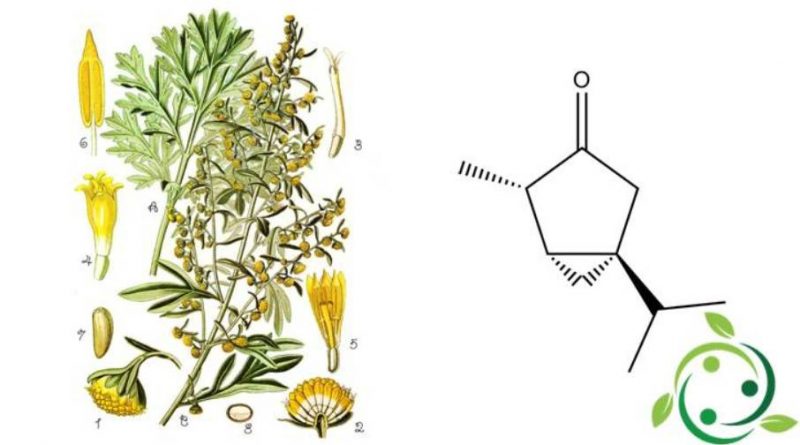
Il tujone o absintolo, il cui termine nella nomenclatura ufficiale IUPAC è: 4R-metil-1S-propan-2-il-5R-biciclo[3.1.0]esan-3-one è un chetone e un terpenoide che esiste in due forme stereoisomere: (+)-3-tujone o α-tujone e (−)-3-tujone o β-tujone.tujone.
Il tujone ha formula bruta o molecolare : C10H16O e presenta un odore di mentolo.
Questo composto si trova in molte specie di assenzio in modeste quantità e nel tanaceto; si trova, inoltre in tracce anche nella salvia, nell’issopo ed in altre piante.
In particolare l’Assenzio è tradizionalmente noto a tutti per le sue caratteristiche spiccatamente amare e per la diffusa credenza che le sue preparazioni sono tossiche. Nel suo olio essenziale è presente, come detto, il tujone, che ad alte dosi può dare seri disturbi; tuttavia, contenendo il consumo alle reali necessità e non superando le dosi indicate, le preparazioni di Assenzio possono essere utilizzate con buona sicurezza ed efficacia; si eviti al più di consumare liquori a base di Assenzio perché risulterebbero particolarmente ricchi di tujone e quindi dannosi alla salute.
Nella medicina popolare l’assenzio è stato utilizzato nella cura dei sintomi dispeptici, oltre che come eupeptico (sostanza di intenso sapore amaro che favorisce la digestione) e carminativo (sostanza che favorisce l’eliminazione di gas dallo stomaco e dall’intestino). L’assenzio possiede, inoltre, proprietà antimicrobiche. In un recente studio è, inoltre, stata osservata la capacità dell’olio essenziale di inibire la crescita di Candida albicans e Saccharomyces cerevisiae.
Tra gli effetti biologici determinati dal tujone si dimostra che questa sostanza è porfirogenica (causa un accumulo di protoporfirina) e può causare porfiria.
L’accumulo di porfirina indotto dal terpene viene ulteriormente incentivato dalla contemporanea somministrazione di desferossamina, un chelante per il ferro che inibisce la biosintesi dell’eme e mima il blocco che si verifica nei casi di porfiria acuta. Da questi studi risulta pertanto che la somministrazione di tujone può essere pericolosa in pazienti con deficit nella biosintesi dell’eme a livello epatico.
La neurotossicità del tujone è stata associata alla sua capacità di bloccare a livello cerebrale i recettori dell’acido γ-amminobutirrico (GABA) e, in particolare, uno studio condotto da Hold e coll. ha dimostrato che il tujone agisce da antagonista dei recettori GABAA.
La riduzione dell’attività gabaergica prodotta dal tujone contenuto nell’assenzio sembra favorire l’insorgenza di scariche elettriche neuronali anomale, responsabili delle manifestazioni cliniche di tipo comiziale; è stato inoltre ipotizzato che l’attività pro-convulsivante dell’α-tujone possa essere correlata anche ad una riduzione della risposta del recettore 5-HT3 per la serotonina.
Tra gli altri effetti legati al tujone, si ricorda che questa sostanza possiede una lieve affinità per i recettori dei cannabinoidi senza tuttavia indurre effetti cannabis-mimetici. La comparsa di effetti tossici è legata comunque alla concentrazione.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.