Bergamotene
Bergamotene
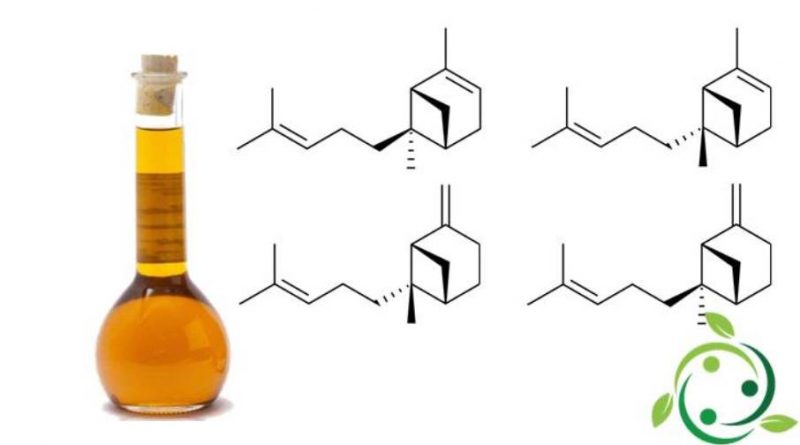
I bergamoteni sono un gruppo di composti chimici con formula bruta o molecolare: C15H24.
Di questi composti esistono due isomeri strutturali, che sono: α-bergamotene e β-bergamotene, che differiscono solo per la posizione di un doppio legame.
Il loro termine nella nomenclatura ufficiale IUPAC è:
– (α): 2,6-dimetil-6- (4-metilpent-3-enil) biciclo [3.1.1] ept-2-ene;
– (β): 6-metil-2-metilidene-6- (4-metilpent-3-en-1-il) biciclo [3.1.1] eptano.
Entrambi questi isomeri hanno, a loro volta degli stereoisomeri, i più comuni dei quali prendono il prefisso di cis e trans.
In natura i bergamoteni si trovano in numerose piante, ed in particolare nei loro oli essenziali.
L’α-Bergamotene si trova negli oli di carota, bergamotto, lime, cedro, semi di cotone, kumquat, basilico, ecc..
I bergamoteni presentano, inoltre, un’altra peculiarità: per alcuni insetti sono feromoni.
Ad esempio, il β- trans -bergamotene è un feromone per la vespa Melittobia digitata .
Le piante, nella loro evoluzione, hanno sviluppato delle forme di difesa agli attacchi degli insetti erbivori producendo feromoni come i bergamoteni che attirano i predatori degli insetti erbivori.
In un meccanismo più complesso, una specie di tabacco, la Nicotiana attenuata (Nicotiana attenuata Torr. ex S.Watson) emette di notte α- trans -bergamotene dai suoi fiori per attirare una falena la falena: la sfinge del tabacco falco di tabacco (Manduca sexta Linnaeus, 1763) come impollinatore; tuttavia, durante il giorno le foglie producono α- trans -bergamotene per attirare gli insetti predatori per nutrirsi di larve e uova che l’impollinatore potrebbe aver prodotto.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.