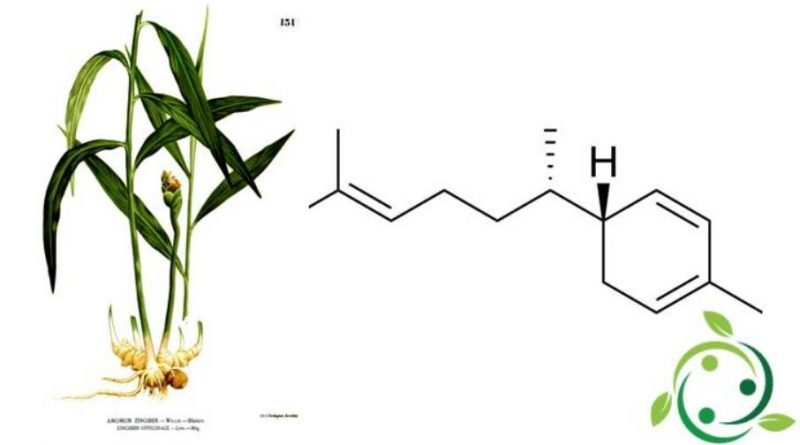
Zingiberene
Zingiberene
Il zingiberene il cui termine nella nomenclatura ufficiale IUPAC è: 2-metil-5- (6-metilept-5-en-2-il) cicloesa-1,3-diene è un sesquiterpene monociclico con formula bruta o molecolare: C15H24.
Lo zingiberene è il costituente predominante dell’olio essenziale dello zenzero (Zingiber officinale Roscoe, 1807) da cui prende il nome.
Questo sesquiterpene può contribuire fino al 30% degli oli essenziali nei rizomi di zenzero e conferisce allo zenzero il suo particolare aroma.
Lo zingiberene, oltre ad essere la molecola più abbondante nell’olio di zenzero è presente in una vasta gamma di specie vegetali, tra cui ricordiamo: pomodoro, basilico, curcuma, cardamomo e sorgo.
La presenza così diffusa di questa molecola in queste piante è probabilmente associata alla sua capacità di difenderle contro gli insetti parassiti; è probabile che questa molecola adempia a vari ruoli tra cui quello di indurre una interruzione nella deposizione delle uova di questi insetti, dissuadendoli, altresì, dal nutrirsi della pianta.
Dal punto di vista biochimico lo zingiberene si forma nella via isoprenoide dal farnesil pirofosfato (FPP).
L’FPP subisce una riconversione per dare origine al nerolidil difosfato. Dopo la rimozione del pirofosfato, l’anello si chiude lasciando un carbocatione sul carbonio terziario attaccato all’anello. Dopo lo spostamento di alcuni idruri si arriva alla formazione dello zingiberene è alla rimozione del protone allilico ciclico e la conseguente formazione di un doppio legame.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.