Bisabolene
Bisabolene
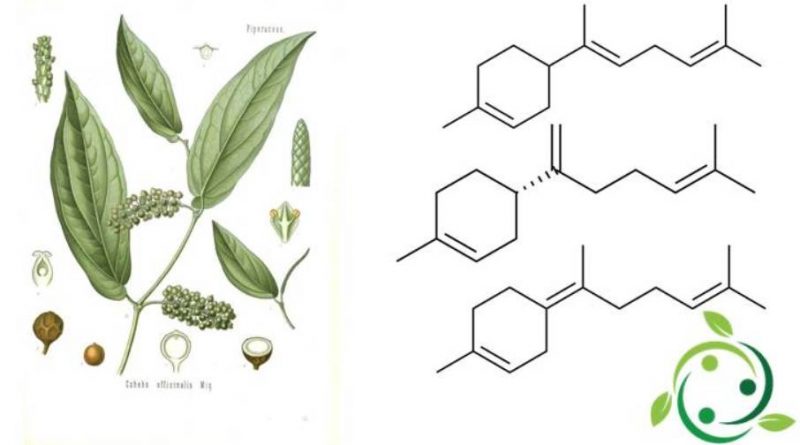
I bisaboleni sono un gruppo di composti chimici naturali strettamente correlati che sono classificati come sesquiterpeni. Nello specifico si hanno tre isomeri, che differiscono per la posizione dei doppi legami carbonio-carbonio, con le configurazioni α, β e γ che prendono rispettivamente nella nomenclatura ufficiale IUPAC i nomi di:
– (α): (E) -1-Metil-4- (6-metilepta-2,5-dien-2-il) cicloes-1-ene;
– (β): (S) -1-Metil-4- (6-metilepta-1,5-dien-2-il) cicloes-1-ene;
– (γ): (Z) -1-Metil-4- (6-metilept-5-en-2-ilidene) cicloes-1-ene.
I bisaboleni hanno formula chimica bruta o moleolare: C15H24 e sono prodotti dal farnesil pirofosfato (FPP) che è un composto intermedio a 15 atomi di carbonio della via metabolica dell’acido mevalonico, utilizzata dagli organismi viventi nella biosintesi dei terpeni, terpenoidi e steroli.
I bisaboleni quindi sono dei composti presenti in natura negli oli essenziali di una grande varietà di piante.
Questi composti sono presenti, tra le altre specie vegetali, nell’angelica (Angelica archangelica L.), nello zenzero (Zingiber officinale Roscoe, 1807) ma anche nel cubebe, nel limone e nell’origano.
Molti derivati del bisabolene agiscono anche come feromoni in diversi insetti, come cimici e moscerini della frutta, inoltre questi composti sono prodotti da diversi funghi, anche se il loro ruolo biologico al momento rimane poco chiaro.
I bisaboleni, inoltre, sono intermedi nella biosintesi di molti altri composti chimici naturali, tra cui l’ernandulcina, un dolcificante naturale.
Il β-bisabolene ha un odore balsamico ed è approvato in Europa come additivo alimentare.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.