Cistina
Cistina
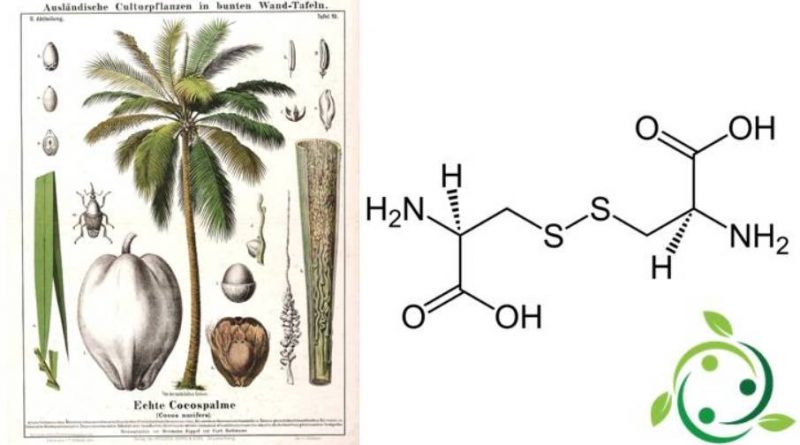
La cistina il cui termine nella nomenclatura ufficiale IUPAC è: acido (2R,7R)-3,3′-ditiobis(2-amminopropanoico), è amminoacido solforato con formula bruta o molecolare: C6H12N2O4S2.
La cistina è quindi un composto chimico solforato costituito dall’interazione dei gruppi SH di due molecole di cisteina e conseguente formazione di un ponte disolfuro S–S.
Si presenta come un solido incolore con punto di fusione 260-261 °C.
Questa molecola fu scoperta nel 1810 da William Hyde Wollaston ma fu riconosciuta come un componente delle proteine solamente quando venne isolata dalle corna di una vacca, nel 1899.
Questa molecola entra nella costituzione di molte proteine. La quantità dei ponti S–S di c. si trova con la massima frequenza nella cheratina, mentre alcune proteine dei Vertebrati ne sembrano del tutto prive, e le proteine globulari, compresi gli enzimi, ne contengono quantità variabili.
La cistina, in genere, nei tessuti degli organismi viventi, è tra gli amminoacidi meno frequenti (1-2% dei residui totali) a eccezione della pelle e dei peli che ne contengono fino al 5%.
La cistina si trova inoltre come amminoacido libero in quasi tutti i fluidi biologici ed è un amminoacido non essenziale per l’uomo e gli animali (può essere sintetizzato dalla metionina). Per riduzione reversibile dà origine a due molecole di cisteina: pertanto i due amminoacidi seguono nell’organismo le stesse vie metaboliche. I legami disolfuro sono in grado di unire catene polipeptidiche diverse o creare ponti tra zone distanti di una stessa catena, determinando così la forma della molecola e la posizione relativa nello spazio dei gruppi che la costituiscono.
In natura la cistina è contenuta in alcuni vegetali come nella Palma da cocco (Cocos nucifera L., 1753), inoltre i supplementi dietetici di N-acetilcisteina sono una fonte di cistina, ma le dosi sono limitate dagli effetti collaterali. Una delle fonti più ricche di cistina è rappresentata dalle proteine non denaturate del siero di latte.
La cistina non viene digerita o idrolizzata in modo significativo nello stomaco, ma viene trasportata dal flusso sanguigno nei vari distretti cellulari dell’organismo. Qui il debole legame disolfuro viene scisso producendo la cisteina, che viene resa disponibile per la sintesi del glutatione.
Questo amminoacido è significativamente presente nei peli umani, in particolare è responsabile della forma ondulata dei capelli, ondulazione dovuta alla formazione dei caratteristici ponti disolfuro (-S-S-).
La cistina viene inoltre utilizzata dall’organismo quale fonte di cisteina necessaria per la biosintesi del glutatione, insieme col glutammato e con la glicina. In particolare, la cistina è ritenuta la forma molecolare preferita dalle cellule del sistema immunitario, inclusi macrofagi e astrociti. I linfociti e i neuroni preferiscono invece utilizzare direttamente la cisteina per effettuare la sintesi del glutatione.
Si ricorda, inoltre, che la cistinosi è una malattia ereditaria connessa a un’alterazione del metabolismo degli amminoacidi e segnatamente della cistina, che si deposita in vari organi e viene abbondantemente eliminata con le urine (cistinuria). La cistina, poco solubile, tende ad accumularsi nelle cellule reticolo-istiocitarie del rene (oltreché del fegato, della milza, dei linfonodi e della congiuntiva), provocando alterazioni della funzione tubulare renale. Se la malattia non è tempestivamente diagnosticata e trattata, la prognosi è severa. La cistinuria si riscontra in vari quadri morbosi e anche in un’altra affezione ereditaria per lo più benigna (a volte addirittura asintomatica), ma talora accompagnata dalla formazione di calcoli renali di cistina.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.