Teobromina
Teobromina
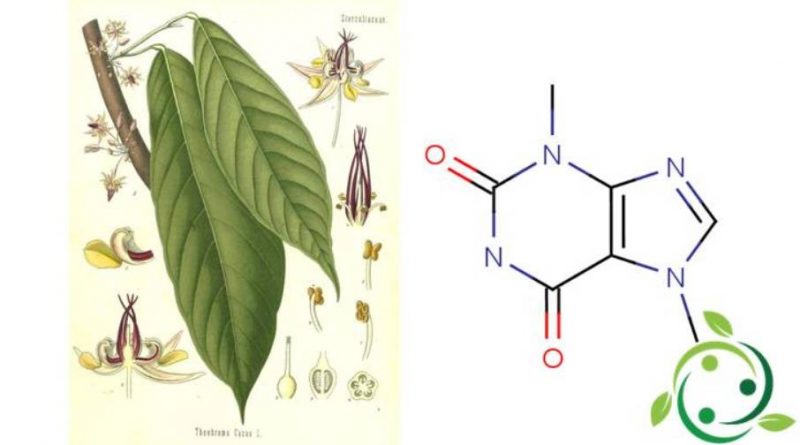
La teobromina, il cui termine nella nomenclatura ufficiale IUPAC è: 3,7-dimetilxantina è un alcaloide con formula bruta o molecolare: C7H8N4O2, che a temperatura ambiente si presenta come un solido bianco.
La teobromina è un composto naturale presente nelle piante di cacao (Theobroma cacao L., 1753) e nei suoi derivati. È anche presente in piccole quantità nelle foglie di tè, nel guaranà, nella cola e nel mate.
Gli effetti stimolanti del cacao sono proprio legati alla presenza di teobromina (contenuta in misura del 2% circa), congiuntamente alla caffeina (0,6-0,8%).
Mediamente, in una barretta al cioccolato fondente da 100 grammi, possiamo ritrovare 600-1800 mg di teobromina e 20-60 mg di caffeina.
Ovviamente si tratta di valori generali, che possono variare – anche considerevolmente – in relazione al tipo di semi, alle tecniche colturali e al processo di fermentazione a cui vengono sottoposti prima di essere torrefatti.
Nel cioccolato al latte il contenuto in teobromina è ridotto (perché minore è la percentuale di cacao), mentre è praticamente nullo in quello bianco.
Questo alcaloide svolge un’azione stimolante del sistema nervoso centrale intervenendo sulle sinapsi; inoltre possiede una lieve azione diuretica e presenta un modesto effetto vasodilatatore.
La teobromina è un ingrediente alquanto comune in alcuni integratori energetici ed in certe creme per il trattamento della cellulite.
La teobromina, quando viene consumata in grandi quantità, è particolarmente tossica per i cani.
L’effetto stimolante sul sistema nervoso centrale è circa 10 volte inferiore rispetto a quello della caffeina; a dosi elevate, comunque, la teobromina può causare irrequietezza, tremori, ansietà, sudori, aritmie, perdita di appetito, nausea e vomito.
Negli uomini, questa sostanza, ha una tossicità trascurabile, dato che viene metabolizzata molto velocemente mentre, come detto, in altri animali, come per esempio cani e gatti, è tossica perché la metabolizzano molto lentamente.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.