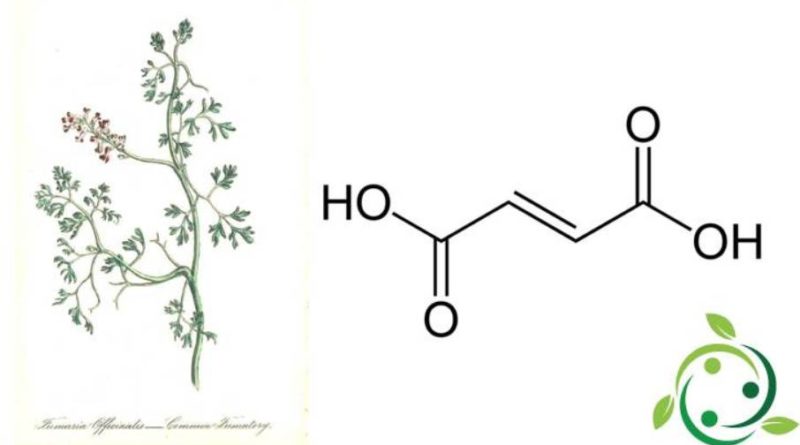
Acido fumarico
Acido fumarico
L’acido fumarico, il cui termine nella nomenclatura ufficiale IUPAC è: acido E-butendioico, ha formula bruta o molecolare: C4H4O4.
L’acido fumarico è un acido presente in natura nella frutta e verdura, ed è stato, inoltre, isolato nelle radici della Fumaria officinalis (Fumariaceae) da cui prende il nome e si trova oltre che nelle piante del genere Fumaria, nei funghi porcini (in particolare nel Boletus fomentarius var. Pseudo-igniarius), nei licheni e nella Cetraria islandica.
Dal punto di vista chimico l’acido fumarico è un isomero dell’acido maleico (acido cis-butendioico); le due sostanze sono isomeri stabili e distinti, dato che la rotazione attorno ad un legame doppio carbonio-carbonio è impedita, in quanto la transizione tra gli isomeri cis (Z) e trans (E) richiede una notevole energia di attivazione.
Pur tuttavia le caratteristiche dei due acidi risultano alquanto diverse in quanto l’acido maleico è tossico, mentre il fumarico è usato come medicinale ed in prodotti alimentari.
Il prodotto commerciale, utilizzato dall’industria alimentare, è ottenuto attraverso la fermentazione di zuccheri (via fermentativa) effettuata da funghi, oppure attraverso la sintesi chimica (via sintetica). Nella via sintetica, l’acido fumarico viene soprattutto prodotto per isomerizzazione dell’acido maleico, utilizzando come catalizzatore l’urea.
In passato l’acido fumarico è stato preparato per la prima volta a partire dall’acido succinico ed una tradizionale via di sintesi prevedeva soprattutto l’ossidazione del furfurale (ottenibile dalla trasformazione del mais) utilizzando un clorato in presenza di un catalizzatore a base di vanadio.
L’acido fumarico viene utilizzato come conservante e come regolatore di acidità e stabilizzante di numerosi cibi, generalmente in sostituzione dell’acido tartarico e a volte, dell’acido citrico ed è classificato come E297.
Oltre che come conservante, viene anche impiegato in medicina per il trattamento della psoriasi, una delle più comuni forme croniche di malattia della pelle.
In questo caso l’acido fumarico viene somministrato solitamente in dosi di 60-105 mg al giorno, che possono anche essere incrementate.
Tra gli effetti collaterali, se usato come medicinale, vi possono essere disordini gastrointestinali e ai reni, o arrossamento della pelle. In tutti gli altri casi, quindi se consumato in quantitativi normali, non sembra produrre effetti collaterali nocivi.
Si ricorda, inoltre che, il fumarato, che è la sua forma anionica, è un intermedio nel ciclo dell’acido citrico utilizzato dalle cellule per produrre energia sotto forma di adenosin trifosfato (ATP) dagli alimenti. Esso è formato dall’ossidazione da parte della succinato deidrogenasi del succinato. Successivamente il fumarato viene convertito dalla fumarasi a malato. La pelle umana produce naturalmente acido fumarico quando questa è esposta alla luce solare.
Tra gli altri utilizzi si ricorda il suo impiego nella sintesi di resine poliestere e polialcoli e come mordente per i coloranti ed, inoltre, utilizzato per sintetizzare una delle prime strutture metallo-organiche che presentano applicazioni commerciali grazie alle sue notevoli proprietà meccaniche e di adsorbimento, in combinazione con una bassa tossicità rispetto ad altri MOFs ben studiati.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.