Arabinosio
Arabinosio
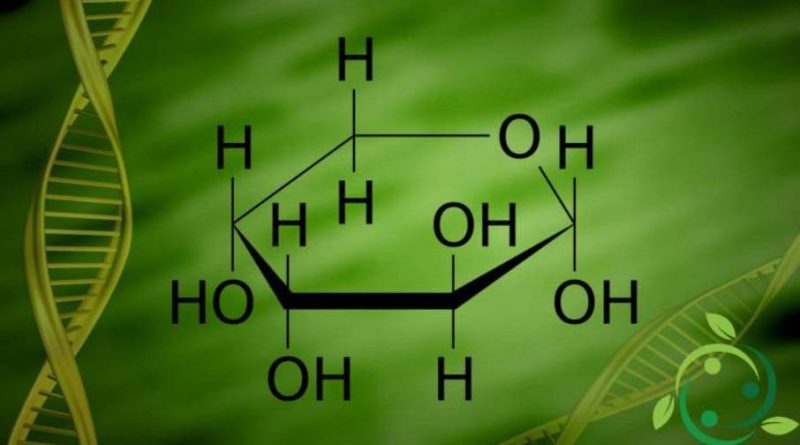
L’arabinosio, la cui formula bruta o molecolare è: C5H10O5 è uno zucchero aldeidico a 5 atomi di carbonio largamente diffuso nel regno vegetale dove è presente sotto forma di complessi polisaccaridici (pentosani) oppure libero, come nel legno di cedro rosso e in varie specie di Pinus.
A temperatura ambiente si presenta come un solido cristallino incolore ed il suo nome deriva dalla gomma arabica, composto da dove venne isolato per la prima volta.
L’arabinosio, è presente nell’uomo ed è un costituente dell’urina, nella quale può raggiungere concentrazioni elevate per effetto di diete ad alto contenuto proteico oppure a causa di una malattia metabolica (pentosuria).
La sintesi biochimica nelle piante prende origine dall’L-ribulosio per azione dell’enzima L-arabinosio-isomerasi. L’L-arabinosio riduce il liquido di Fehling e si trasforma a caldo in furfurale. L’isomero destrogiro, D-arabinosio, è presente nei micobatteri e in alcuni glucosidi (aloina). L’L-arabinosio trova impiego in microbiologia per la preparazione di terreni di coltura batterici.
L’arabinosio, a differenza di molti altri saccaridi, si trova negli organismi prevalentemente nella sua configurazione L (levogira) piuttosto che in quella D (destrogira) ed in natura lo si può trovare nei tessuti vegetali sotto forma di saponina, o come componente di biopolimeri come l’emicellulosa e la pectina.
Una importante applicazione in campo alimentare dell’arabinosio è quella che trova alcuni impieghi soprattutto come dolcificante, consigliato per i pre-diabetici ed i diabetici, essendo un antagonista della saccarasi, l’enzima che scinde fruttosio e glucosio nell’intestino crasso.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.