Acido barbiturico
Acido barbiturico
L’ acido barbiturico, il cui termine nella nomenclatura ufficiale IUPAC è: 1H,3H,5H-pirimidin-2,4,6-trione, ed il cui nome alternativo è: malonilurea 2,4,6-triidrossipirimidina è un acido organico avente formula bruta o molecolare: C4H4N2O3.
Dal punto di vista chimico l’acido barbiturico è l’ureide ciclica dell’acido malonico.
Il nome venne coniato dal chimico tedesco A. Baeyer che nel 1863 preparò questo composto scaldando l’acido malonico (estratto dalla barbabietola) con urea.
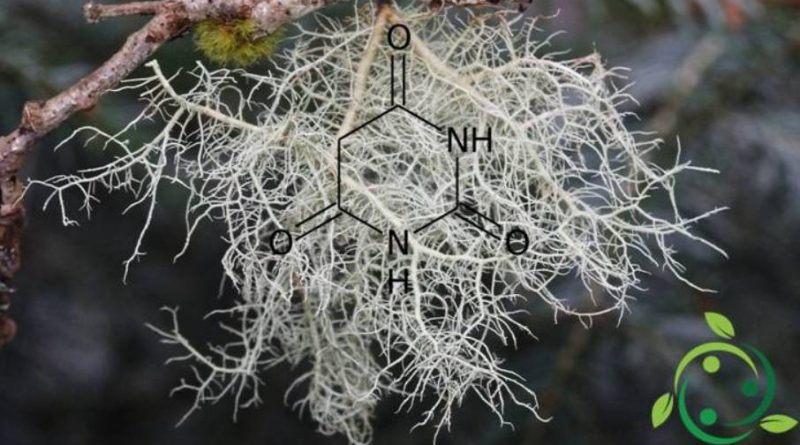
Dall’acido barbiturico derivano una importante serie di farmaci noti con i nomi di barbiturici. Il termine barbiturico deriva dal lichene Usnea barbata (barba di bosco) da cui si ricava l’acido malonico, e dall’urea.
L’ acido barbiturico a temperatura ambiente si presenta come un solido biancastro e con un odore tenue.
La categoria dei barbiturici era molto utilizzata in passato in terapie cliniche. Molti suoi derivati sono usati come sonniferi.
Altri derivati, contenenti zolfo e detti tiobarbiturici, sono caratterizzati da un’azione ipnotica particolarmente intensa, rapida e breve e vengono pertanto adoperati come anestetici, per via endovenosa. A dosi alte i barbiturici danno fenomeni tossici (barbiturismo) seguiti spesso da morte.
Così, ben presto, ci si accorse che questa categoria di prodotti causavano dipendenza e risultavano, inoltre, particolarmente pericolosi.
Così negli anni settanta del secolo scorso ben presto furono sostituiti dalle benzodiazepine.
I derivati dell’ acido barbiturico, seppur in disuso, oggi continuano ad essere prescritti come antiepilettici e per il trattamento di eventi traumatici della testa, inoltre sono utilizzati anche in anestesia.
Da precisare che l’acido barbiturico è tossico ma, a differenza dei suoi derivati, non risulta farmacologicamente attivo.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.