Acido glucuronico
Acido glucuronico
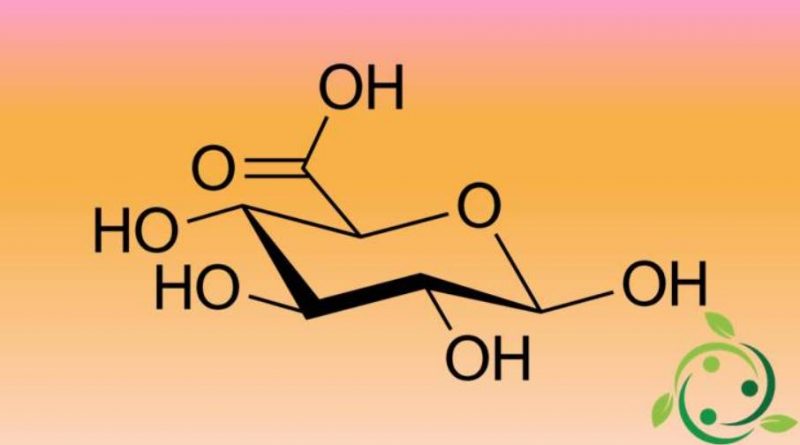
L’ acido glucuronico (più precisamente acido D-glucuronico), la cui formula bruta o molecolare è: C6H10O7, è un acido organico della famiglia degli acidi alduronici. La forma levogira dell’ acido glucuronico è l’ acido iduronico (acido L-iduronico).
L’ acido glucuronico viene prodotto dall’ossidazione del gruppo alcolico primario (l’OH legato al C-6) del D-glucosio a gruppo carbossilico.
All’interno della sua molecola l’acido glucuronico ha i tre gruppi funzionali differenti: OH, -CHO, -COOH, per cui è un aldossiacido.
L’importanza biologica dell’acido glucoronico è da identificare nei meccanismi di detossificazione operati all’interno delle cellula animali.
La glucuronicazione è il processo di legame di una molecola, generalmente tossica, o xenobiotica ad uno dei gruppi alcolici della molecola. Molte cellule, specialmente gli epatociti, sfruttano questo meccanismo per rendere più solubili le sostanze estranee, ad esempio antibiotici o piccole tossine, e espellerli mediante la funzionalità renale.
A livello del metabolismo gli ormoni steroidei (insolubili in acqua) si trovano nel sangue e nelle urine prevalentemente come glucuronidi (glucosidi dell’acido glucuronico). Anche il fenolo, tossico per l’uomo, viene eliminato come glucuronide per via della maggiore solubilità in acqua di quest’ultimo.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.

Dove si può comprare l acido glicuronico
Buongiorno, trattandosi di prodotti specifici può provare presso i fornitori di reagenti e sostanze chimiche per i laboratori di analisi, oppure può fare una ricerca on-line.