Borneolo
Borneolo
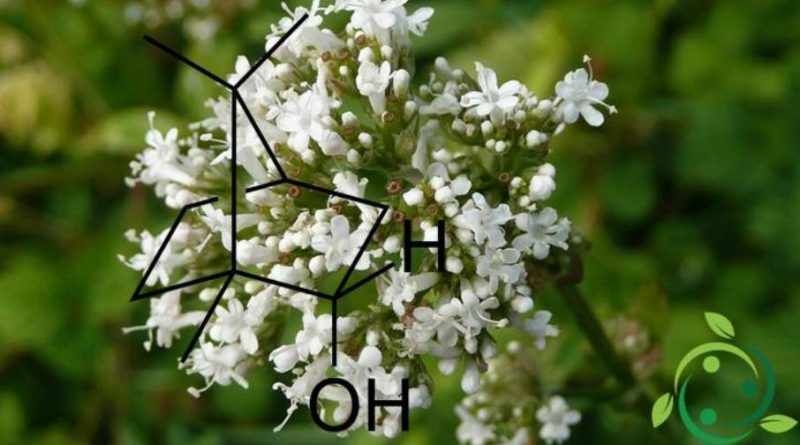
Il borneolo, il cui termine nella nomenclatura IUPAC è: endo-1,7,7-Trimetil-biciclo[2.2.1]eptan-2-olo e la cui formula bruta o molecolare è: C10H18O, è un composto organico biciclico della famiglia dei terpeni. Nel borneolo il gruppo ossidrilico è sistemato in posizione endo.
Vi sono due isomeri ottici di questa molecola dei quali il predominante è la forma d. L’isoborneolo è il corrispondente isomero. L’enantiomero presente in natura è quello destrogiro.
Dall’ossidazione del borneolo si ottiene il corrispondente chetone, la canfora, nota con uno dei suoi nomi storici è cioè: “canfora del Borneo”.
In natura il borneolo è presente in molte specie vegetali tra cui: Teucrium chamaedrys, Satureja montana, Juniperus communis, Cannabis sativa, Coriandrum sativum, Tanacetum vulgare, Rosmarinus officinalis, Centranthus ruber, Achillea millefolium, Artemisia arborescens, Valeriana officinalis, nelle piante del genere Cymbopogon (Citronella) ed alcuni loro rispettivi oli essenziali.
Inoltre, tra le sue caratteristiche interessanti ricordiamo che il borneolo è un repellente per gli insetti.
In laboratorio il borneolo può essere sintetizzato per riduzione della canfora secondo la reazione di Meerwein-Ponndorf-Verley. Dalla riduzione della canfora con sodio boroidruro si ottiene invece l’isoborneolo.
Il borneolo trova impiego nella medicina tradizionale cinese. Viene anticamente menzionato nel Bencao Gangmu, il trattato di erboristeria cinese scritto da Li Shizhen durante la dinastia Ming.
L’assunzione inappropriata di borneolo può causare nausea, confusione, vertigine e convulsioni.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.