Saponine
Saponine
Le saponine (o saponosidi) dal punto di vista chimico sono dei glicosidi terpenici di origine vegetale.
Queste molecole prendono il nome dalla Saponaria officinalis, che veniva coltivata un tempo per il lavaggio della lana.
Le saponine però sono contenute in molte piante ed in alcuni casi la loro presenza può raggiungere anche il 30% del peso secco della pianta.
La caratteristica fisica principale delle saponine è quella di abbassare la tensione superficiale in soluzioni acquose; formano soluzioni colloidali schiumeggianti e si possono usare, quindi, come emulsionanti.
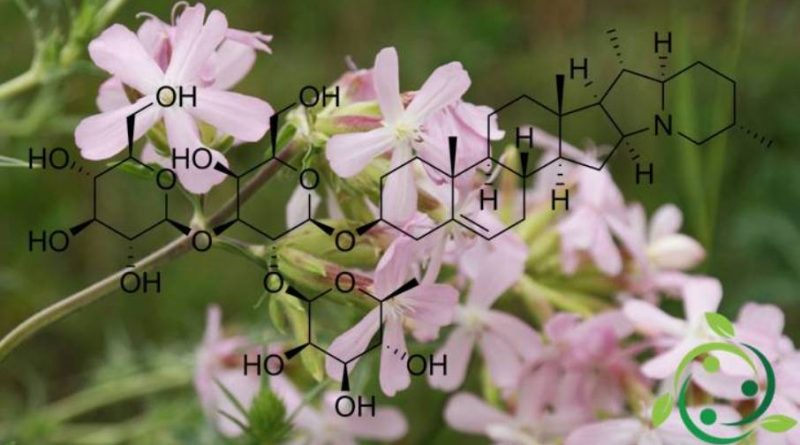
Le saponine sono formate dall’unione di residui zuccherini (quali glucosio, fruttosio, galattosio, arabinosio ed altri) con una molecola non zuccherina (aglicone). Questa struttura molecolare particolare è responsabile dell’attività detergente delle saponine in quanto gli zuccheri formano una sezione idrosolubile mentre l’aglicone risulta essere liposolubile.
Le saponine a nucleo triterpenico (come la glicirrizina della liquirizia o l’escina dell’ippocastano) hanno una struttura pentaciclica a 30 atomi di carbonio mentre quelle a nucleo steroideo ne hanno una di base con 27 carboni (spesso la catena alifatica è trasformata in due anelli eterociclici).
La presenza delle saponine nelle piante pare sia collegata a funzioni difensive contro organismi patogeni, in particolare dei funghi. In alcuni casi le saponine sono già presenti nelle piante; in determinate circostanze, in cui la pianta subisca dei danni, vengono sintetizzate da dei precursori. Tale funzione è avvalorata dal fatto che tra i cereali l’unico che contiene saponine è l’avena; questa pianta infatti risulta il più resistente fra i cereali alle muffe ed ai funghi.
È stato accertato che il meccanismo di azione fungicida sia dovuto alla reazione delle saponine con steroli presenti nelle membrane delle ife dei funghi, alla rottura della membrana ed alla conseguente morte del fungo.
Le saponine sono molecole con proprietà chimico – fisiche molto importanti soprattutto per l’industria cosmetica e dermo – funzionale, dove vengono utilizzate come veicolanti.
Infatti il nucleo triterpenico, così come quello steroidico, è facilmente individuabile come porzione di molecola lipofila, poco solubile in acqua; la sua glicosilazione ne permette invece la solubilità. Questa duplice caratterizzazione: aglicone lipofilo e glicone idrofilo, rende la molecola anfifilica (o anfipatica), cioè con un polo affine all’acqua e l’altro affine a sostanze oleose e grasse, quindi lipofile.
Per questa loro peculiarità quindi, le saponine, grazie a questa importante particolarità, sono utilizzate come veicolanti di molecole idrofile su una matrice grassa, come potrebbe essere per esempio la cute; si possono trascinare dietro il principio attivo idrofilo, facendolo passare facilmente attraverso una matrice grassa, come quella che costituisce le estremità delle membrane cellulari. Questo accade anche per prodotti rassodanti e anticellulite contenenti caffeina, principio attivo idrofilo trascinato dalle saponine nelle parti più profonde del derma.
Le saponine sono presenti in differenti droghe con differenti profili d’impiego e, fondamentalmente, le saponine hanno un’azione irritante su tutte le mucose; azione che è proporzionale alla dose impiegata. La proprietà espettorante della glicirrizina (contenuta nella Liquirizia), è dovuta proprio all’azione irritante che questa molecola determina sui bronchi; a sua volta, l’azione irritante determina la secrezione di un muco più fluido, che favorisce l’eliminazione di quello più denso congestionante.
Altra proprietà generalmente ascrivibile alle saponine, è quella capillarotropa, ovvero capace di agire direttamente sui capillari aumentandone la resistenza e diminuendone la permeabilità . Hanno anche azione sui vasi della muscolatura liscia, dove determinano vasocostrizione; aumentano altresì la permeabilità delle membrane, causando uno scompenso strutturale del mosaico fluido e delle proteine di membrana che facilitano il trasporto di svariate sostanze come possono essere i principi attivi.
In definitiva, in terapia, al momento, alle saponine di alcune piante viene riconosciuta attività antinfiammatoria, cicatrizzante (come nella liquirizia) ed antiedemigena (come nell’ippocastano).
Spesso le saponine sono usate nell’industria per la successiva produzione di ormoni steroidei (come, ad esempio, il testosterone ed il cortisolo).
Si ricorda che l’iniezione per via parenterale di questi composti determina emolisi.
L’assunzione per via orale, invece, non produce quest’effetto velenoso in quanto l’attività emolitica delle saponine si esplica solo se la molecola rimane intatta, cosa che durante la digestione non avviene in quanto essa viene scissa nei due suoi componenti.
In animali a sangue freddo e nei pesci le saponine risultano tossiche in ogni caso.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.