Acido umico
Acido umico
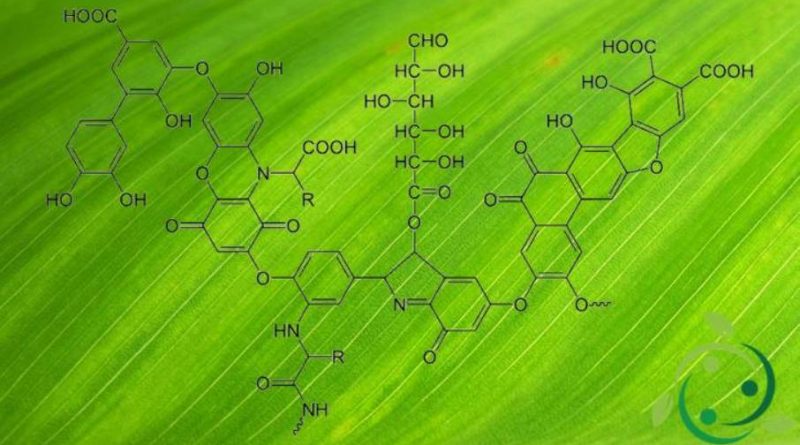
Gli acidi umici, in inglese Humic Acids (HAs), sono delle sostanze di origine naturale provenienti della biodegradazione microbica di sostanze organiche, sia di natura vegetale che animale. Sotto la comune nomenclatura di acido umico rientra una miscela di diversi acidi organici contenenti gruppi carbossilici e fenolici. Le sostanze umiche possono a loro volta suddividersi in tre differenti categorie: umina, che è la componente legata alla matrice minerale, gli acidi umici veri e propri e gliacidi fulvici, in funzione della loro solubilità in acqua a a differenti valori del pH. A differenza dell’acido fulvico l’acido umico è insolubile in acqua a pH ≤ 1.
Di fatto l’acido umico è un biopolimero ma non costituisce un composto ben definito; la struttura degli acidi umici infatti varia a secondo della sostanza di provenienza, dell’ecosistema e dell’habitat specifico di riferimento. La presenza degli acidi umici è maggiore nei suoli fertili e nella soluzione circolante dei suoli; sono componenti tipici dell’humus che è il fertilizzante naturale proveniente dalla biodegradazione dei residui vegetali ed animali in determinate condizioni di aerazione ed umidità.
L’importanza dell’acido umico nel terreno è legata alle sue particolari funzioni; gli acidi umidi del suolo svolgono reazione di ossidoriduzione, funzioni tampone, di ritenzione idrica, formano legami con ioni metallici, assorbono soluti organici, svolgono azione biostimolante nei delle piante e contribuiscono alla degradazione naturale degli inquinanti tossici.