Carvone
Carvone
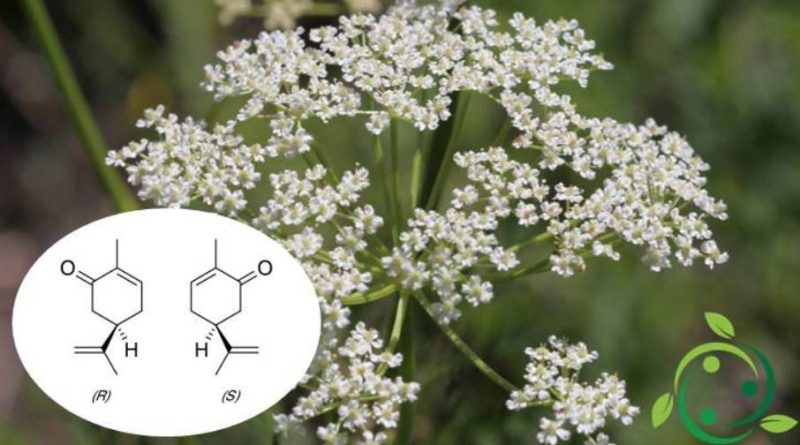
Il carvone il cui termine nella nomenclatura ufficiale IUPAC è: (R)-5-Isopropenil-2-metil-2-cicloesenone ha formula bruta o molecolare: C10H14O.
Il carvone, dal punto di vista della classificazione chimica è un terpenoide con punto di ebollizione a 231 °C.
Questa molecola è solubile in alcol e cloroformio ed è composta da due enantiomeri entrambi presenti in natura; lo si ritrova infatti, in particolar modo nell’olio dei semi di cumino dei prati (Carum carvi), del mentastro verde (Mentha spicata) e dell’aneto.
In natura l’S-(+)-carvone è il principale costituente (60-70%) dell’olio di semi di cumino (Carum carvi). È comunque presente intorno al 40-60% nell’olio di semi di aneto (Anethum graveolens), e anche nell’olio di buccia d’arancia.
L’R-(-)-carvone è invece il componente predominante dell’olio essenziale di diverse specie di menta, in particolare dell’olio di menta (Mentha spicata), intorno al 50-80% di R-(-)-carvone.
Anche se la menta è una ottima fonte naturale di questo composto, la maggior parte di R-(-)-carvone presente in commercio è sintetizzato a partire dall’ R-(+)-limonene.
L’R-(-)-Carvone si trova anche nell’olio di kuromoji (Lindera Umbellata). Alcuni oli, come l’olio della pianta di zenzero, contengono una miscela di entrambi gli enantiomeri.
Molti altri oli naturali, per esempio l’olio di menta piperita, contengono tracce di carvoni.
Comunque sia entrambi i carvoni sono utilizzati nell’industria alimentare come aromi. L’ R-(−)-Carvone viene usato anche come essenza per rinfrescare l’aria e, come molti oli essenziali, gli oli contenenti carvone sono usati nella aromaterapia e nella medicina alternativa.
Per le sue peculiarità è utilizzato per la creazione di profumi ed aromi.
I recettori olfattivi percepiscono i due enantiomeri in maniera differente. L’R-(–)-carvone, che profuma come foglie di menta, mentre l’S-(+)-carvone, che emette un odore tipico dei semi di cumino.
Le peculiarità del carvone, pur se non conosciuto come tale, sono stat sfruttate dall’natichita tramite la pianta del cumino dei prati.
Questa specie è stata utilizzato per scopi medicinali dagli antichi romani.
Solo nel 1849, ad opera di Franz Varrentrapp (1815-1877), il carvone è stato isolato come un composto puro.
Oltre alle già citate peculiarità del carvone questa molecola trova impiego in molti campi ed applicazioni.
Infatti essendo questa molecola responsabile del profumo del cumino, dell’aneto e della menta, è usata da millenni per dare particolare aroma ai cibi.
In agricoltura, inoltre, l’S-(+)-Carvone è usato per prevenire il germoglia mento delle patate durante il periodo di stoccaggio.
L’altro enantiometro, l’ (R)-(–)-Carvone, dopo alcune ricerche, dimostrerebbe attività repellente contro le zanzare.
Avvertenza: le informazioni riportate non sono consigli medici e potrebbero non essere accurate. I contenuti hanno solo fine illustrativo e non sostituiscono il parere medico.