L’Acido citrico
L’Acido citrico
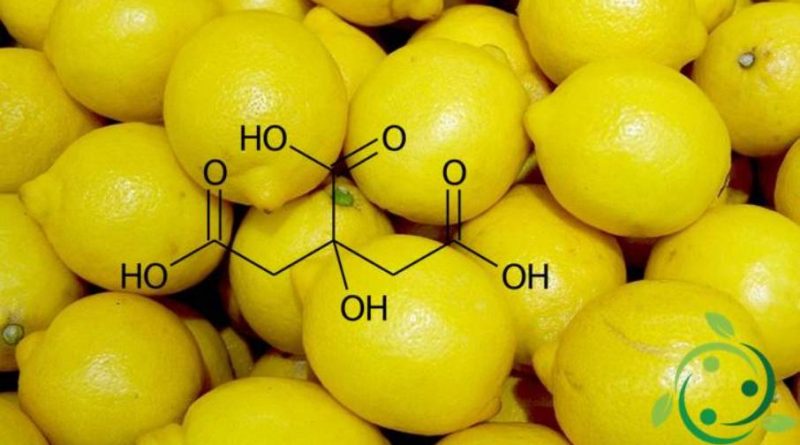
L’Acido citrico, il cui nome nella nomenclatura IUPAC è : acido 2-idrossi-1,2,3-propantricarbossilico e la cui formula bruta è: C6H8O7 è un acido tricarbossilico, solido, incolore, solubile in acqua in un ampio intervallo di pH.
L’acido citrico si trova in tracce nella frutta e soprattutto del genere Citrus. Nel succo di limone la sua presenza è intorno al 3-4% e nell’arancia l’1%. Lo si ritrova anche nel legno verde, nei funghi, nel tabacco, nel vino e persino nel latte. Dall’acido citrico, tramite reazioni con composti basici si ottengono i Sali corrispondenti che sono i citrati.
La presenza dell’acido citrico in natura è notevole; oltre alle verdure e alla frutta le piante che contengono la maggior concentrazione di acido citrico sono gli agrumi e tra questi limoni e lime che hanno concentrazioni particolarmente elevate dell’acido.
Fu nel 1784, ad opera di Carl Wilhelm Scheele, che venne isolato per la prima volta l’acido citrico dal succo di limone tramite cristallizzazione. Bisogna aspettare il 1890 per avere la produzione a livello industriale con estrazione chimica dal succo di limone. Successivamente si scopri che sia il Penicillium che alcuni ceppi di Aspergillus niger potevano sovraintendere alla trasformazione di sostanze zuccherine in acido citrico.
Oggi l’acido citrico viene prodotto attraverso fermentazioni su scala industriale utilizzando funghi (come l’Aspergillus niger) o lieviti in bioreattori ad ambiente a basso pH e bassa concentrazione di ferro.
L’acido citrico è un importante prodotto intermedio nel catabolismo dei carboidrati, una via metabolica fondamentale per gli esseri viventi aerobici, sia piante, che batteri e animali, incluso l’uomo. Il relativo processo biochimico prende il nome di ciclo di Krebs.
L’acido citrico è considerato un fattore nutrizionale. Non è essenziale, poiché l’organismo è in grado di produrlo autonomamente, ma esercita comunque un ruolo molto importante per le funzioni biologiche. Le sue funzioni principali sono: Assieme alla vitamina C (acido ascorbico) favorisce l’assorbimento del ferro; ostacola la formazione di calcoli renali di cistina, xantine e acido urico; è un ottimo alcalinizzante urinario e può essere utilizzato per potenziare l’attività terapeutica di farmaci o integratori che richiedono urine basiche per espletare al meglio la loro azione terapeutica, per favorire l’escrezione di farmaci con caratteristiche di acidi deboli. Espleta inoltre una blanda azione battericida e antiartritica.
L’acido citrico ha molteplici usi ed applicazioni:
– ha la proprietà di ridurre la durezza dell’acqua, e per la sua azione anticalcare viene utilizzato in ammorbidenti eco-sostenibili per il lavaggio in lavatrice e come disincrostante per le tubazioni;
– nella cosmesi può essere utilizzato come correttore di pH;
– per le sue capacità disincrostanti e corrosive è ampiamente utilizzato in prodotti per la pulizia di ambienti industriali professionali.
– è utilizzato come aroma e un conservante nei cibi e nelle bevande, specialmente nelle bibite e nelle caramelle;
– nell’industria alimentare è usato, con la denominazione E330, come acidulante e per correggere il pH di coloranti basici, come emulsionante, ad esempio nei gelati, come sostituto del succo di limone oppure per evitare la precipitazione dello zucchero nel caramello;l’acido citrico è molto usato anche in bibite, prodotti di confetteria, gelatine, marmellate, birre e, congiuntamente al sodio bicarbonato, in preparazioni effervescenti.
L’acido citrico trova anche impiego in farmacologia come anticoagulante nella conservazione del sangue estratto e come conservante in preparati farmaceutici. I sali di citrato dei metalli sono usati per fornire i minerali in forma biologicamente più disponibile in molti integratori alimentari.
L’acido citrico, allo stato puro, è irritante per contatto con la pelle e gli occhi per cui va utilizzato abbigliamento protettivo quando si manipola. Un consumo eccessivo, allo stato concentrato, può intaccare lo smalto dentale.